Acrylic acid didn’t always occupy the prominent position it holds today in industry. Looking back to the 19th century, a German chemist named Charles Gerhardt figured out how to bring it into existence by treating ethylene with oxidants. That process laid the groundwork for more efficient ways to make the chemical, paving the way for large-scale production. Early uses started to appear in the textile and adhesives world. Over the decades, manufacturing moved from using acetylene to a safer and more cost-effective technique centered around propylene oxidation. This shift made acrylic acid widely available and affordable. It's hard to ignore the impact this rollout had on everything from diapers to paints. Companies around the world recognized its value, and innovation in its production keeps chugging along even now, due in part to the ever-growing demand for more sustainable and safer synthetic materials.
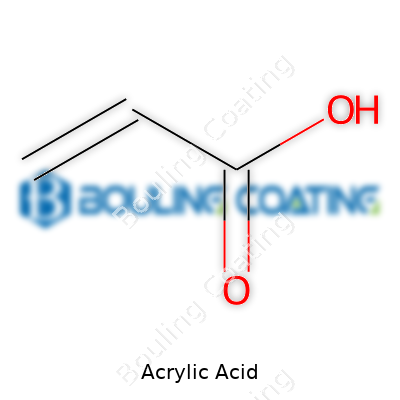
Acrylic acid belongs to the simplest unsaturated carboxylic acids. Its structure seems straightforward, with a double bond between two carbons at center stage, making it reactive and useful in countless applications. Common synonymous names include propenoic acid, 2-propenoic acid, and vinyl formic acid, though most folks in the chemical industry stick to the familiar “acrylic acid.” Its functionality as a monomer gives it the flexibility to form long polymer chains, serving as a building block for synthetic resins and superabsorbent polymers. Business and research sectors lean on this molecule for its reactivity and its ability to establish bonds with a host of other substances, leading to innovations in packaging, water treatment, and even medical materials.
Handling acrylic acid puts you face to face with a clear, colorless liquid with a sharp odor. It boils near 141°C and freezes just below room temperature, making storage and transport somewhat tricky in colder climates. Volatility and a tendency to polymerize on its own can catch producers off guard if stabilizers aren’t added. Its density sits slightly above that of water, and it dissolves readily in alcohols, ethers, and water. On the chemical side, the carboxylic group and the carbon-carbon double bond give it both acidic and reactive characteristics. This means it can link up in countless polymerization and addition reactions, and it often acts as a parent compound for derivatives like esters—serving more end-uses than some give it credit for.
Factories bring acrylic acid to market with varying levels of purity, but the bulk of sales go to users who need it above 99%. Impurities like inhibitors, which prevent pre-mature polymerization, must be listed on any documents to keep buyers informed. Storage requires clear labels warning against sources of ignition and strong bases or oxidizers. Safety data sheets often look intimidating, but they reflect real dangers—acrylic acid can corrode skin and cause nasty fumes if mishandled. Correct labeling helps workers avoid accidental mixing or exposure. Proper packaging, often using coated steel drums with vented closures, prevents self-polymerization and all the headaches that come from an unexpected runaway reaction.
These days, if you follow acrylic acid back to its source, you’ll find propylene at the beginning of most production pipelines. Oxidizing this petrochemical, often in tandem with specialized metal catalyst beds under controlled heat, churns out acrylic acid at scale. Producers work hard to optimize yield, as side reactions can chew up profits and create more byproducts than anyone wants to deal with. The process, once set up, runs continuously and feeds directly into purification columns. Distillation removes water and leftovers, bringing the concentration close to ideal. Some labs still use smaller routes, oxidizing acrolein, but global trends favor propylene-based plants—efficiency, safety, and supply reliability win out in most regions.
Acrylic acid’s unsaturated bond and acidic group invite all kinds of reactions. It loves to polymerize, especially in the presence of free-radical initiators. This trait gets used in manufacturing superabsorbent polymers, acrylate esters, and adhesives. It also forms copolymers with substances like acrylamide, expanding its reach into flocculants and water purification agents. On the organic synthesis front, chemists target both bonds—addition reactions hook new groups onto the double bond, and esterification uses alcohols to form acrylate esters. Its chemical flexibility drives research for new coatings, specialty plastics, even drug delivery devices.
Walk through warehouses and you’ll see labels sporting names like propenoic acid or 2-propenoic acid; some producers tack on terms like “glacial” to indicate high purity levels. Trade names reflect branding more than chemistry, but product sheets nearly always include the IUPAC name or the familiar acrylic acid label. In regulatory filings or customs documents, the CAS number 79-10-7 helps cut through any confusion, ensuring everyone’s speaking the same language.
Handling acrylic acid requires real respect. Its fumes can irritate the eyes, nose, and throat, entering the body through inhalation or skin contact. It causes burns, and repeated exposure leads to chronic health issues. Facilities enforce strict engineering controls—ventilation, containment, and spill response. Workers wear gloves, goggles, and protective clothing—not negotiable. Regular training makes a difference. In the event of a spill, crews move fast with absorbent materials, keeping acid from reaching drains; some companies invest in on-site neutralization technology. Regulations keep getting tighter, with workplace exposure limits reflecting the latest in toxicity research. Safety practices become a way of life when you spend your days around a substance this reactive.
Acrylic acid quietly transforms modern life. In baby diapers and adult incontinence products, it’s the unsung hero behind the superabsorbent materials capturing moisture without leakages. The paints you roll on your house contain acrylic resins—flexible, durable, and weather-resistant. Adhesive manufacturers blend it into tapes and glues for lasting tackiness. Water treatment facilities dose it into flocculants, helping clear out particulates that would otherwise clog up pipelines. Textile plants use it to create wrinkle-resistant finishes, while paper mills rely on it for coatings that boost glossy appearance and strength. Medical suppliers develop skin-adhering gels and wound dressings rolling out of research labs, all based on acrylic acid chemistry.
Chemists keep finding new ways to push acrylic acid further. Many universities and private firms focus on greener synthesis—using bio-based propylene or turning to sugars and glycerol as feedstocks. Recent studies aim to upcycle waste streams or even capture CO₂, turning it into valuable chemicals like acrylic acid. Polymer scientists tweak the monomer’s reactivity, crafting new copolymers for responsive coatings, conductive films, or flexible electronics. In biomedical circles, researchers design hydrogels with tunable properties for controlled drug release, opening new therapeutic doors. Collaboration between academia and industry drives incremental improvements in safety, yield, and downstream product performance.
Acrylic acid’s toxicology has been scrutinized for decades. Acute exposure attacks human skin and mucous membranes, while longer contact ramps up irritation risks. Animal studies raised alarms about reproductive and developmental impacts, guiding regulations on workplace limits. Recent research continues to probe the question of long-term exposure risks, with regulatory bodies like the EPA and OSHA routinely updating guidelines. Environmental analysis of effluents and accidental releases drives stricter numerical limits and best practices for containment. The need for vigilance remains, especially as demand for the chemical rises worldwide. Ongoing toxicology research informs both process safety and environmental protection strategies.
Acrylic acid won’t lose relevance any time soon. The growing thirst for eco-friendly polymers and water-absorbent materials keeps research and production humming. Producers look to slash carbon intensity by transitioning to renewable feedstocks. Bio-based acrylic acid offers a path forward, with pilot plants already achieving decent yields. Advances in catalysis promise fewer byproducts, safer operations, and higher efficiency. New applications appear on the horizon—innovative gels for agriculture, coatings for solar panels, and composite materials linking light weight to toughness. The industry faces calls for better waste management and recycling, nudging both old and new players to revisit processes that date back decades. As consumers keep demanding cleaner, more durable products, acrylic acid stands ready to stand at the center of an evolving world of materials.
Walk down any grocery store aisle or step into a factory and there’s a good chance something around you relied on acrylic acid along the way. A colorless liquid with a sharp scent, this compound shapes dozens of products that make life easier and safer. As someone who has kept an eye on how chemicals shape manufacturing and daily life, I see acrylic acid touching all sorts of sectors without drawing much attention to itself. Yet when digging deeper, its impact becomes clear.
Few people realize how much comfort acrylic acid brings to modern living. Open a baby’s diaper, check a box of incontinence products, or grab a sanitary pad, and you’re dealing with superabsorbent polymers based on polyacrylic acid. This material locks away many times its weight in liquid, which means better hygiene, less odor, and fewer worries for caregivers. In fact, reports show over half the world’s acrylic acid goes into these applications. Wastewater plants also use similar polymers for spills and cleanup—another case where this chemical helps keep public spaces sanitary.
Acrylic acid isn’t just about absorption. Its structure helps build the backbone of paints and coatings. If you ever painted a fence or coated your house, there’s a good chance you used a product carrying acrylic acid derivatives. These resins stick better and fight off sun or rain longer compared to older formulas. That translates to fewer repaint jobs and longer-lasting protection for homes, factories, and public buildings. Plastics and adhesives follow a similar story. Toothpaste tubes, traffic signs, and the glue on sticky notes often owe their stickiness or flexibility to compounds that start with acrylic acid. This versatility comes from the way acrylic acid lets manufacturers tailor molecules for different kinds of toughness and flexibility.
Polyacrylic acid and its cousins show up in textile finishing, lending fabrics softness or helping dyes cling more permanently. There’s a personal memory attached to this: I used to wonder why some clothes stayed colorful wash after wash, and it turns out advancements in finishing chemicals, including acrylic derivatives, made that possible. In water treatment facilities, acrylic acid polymers trap metals or suspend solid waste so that treatment becomes more efficient and water runs cleaner. This directly improves health outcomes and boosts the reliability of municipal water supplies.
There’s always a catch with wide industrial footprints. The production and handling of acrylic acid carry risks: accidental releases or poor safety practices can harm workers or local air quality. Chronic exposure at high levels also raises concerns, so plant managers and governments keep regulations tight. Switching to more sustainable processes, like bio-based routes for acrylic acid, shows promise. Companies in North America and Asia have rolled out such technology, trying to cut down on fossil fuel use and lower carbon footprints. Investing in stronger chemical recycling streams and safer workplace practices also lessens these hazards.
While it’s easy to overlook acrylic acid’s presence, its fingerprints show up in cleaner, safer, and more convenient lives. Paying attention to safer manufacturing and greener sourcing can help keep its benefits rolling without the environmental side effects that sometimes come with heavy industry. As more industries look for ways to balance performance and sustainability, acrylic acid’s story shows how one material can push innovation and caution side by side.
Plenty of industries rely on acrylic acid. People use it to produce plastics, coatings, and even diapers. Despite its usefulness, this chemical is nothing to take lightly. My first brush with a drum of acrylic acid came years ago on a factory floor, where a small spill sent coworkers scrambling to pull on their protective gear. Those moments stick with you, not because of any dramatic outcome, but because they serve as a wake-up call—the stuff isn’t just another industrial fluid.
Acrylic acid can cause burns where it touches skin. Breathing in vapor or mist brings on coughing or shortness of breath, and if it splashes near your eyes, damage follows quickly. Some folks learn about chemical safety the hard way, but experience and available facts reinforce what safety data sheets and warning labels keep repeating: contact is dangerous.
I remember a supervisor’s story from his earlier career, relaying how, after a minor leak, he washed his skin but still developed redness and peeling. It hammered home the need for more than just a casual rinse. Strict policies around acrylic acid don’t just exist to fill manuals. They’re answers to all those hard-earned lessons collected over decades, and they stick around because they keep working.
Start with protective clothing—long sleeves, splash-resistant aprons, and sturdy gloves made from nitrile, polyethylene, or other chemical-resistant materials. Watching coworkers swap out torn gloves opened my eyes to the limits of basic lab gear. Eyes deserve proper goggles, not just glasses, and a face shield if things get splashy. Good ventilation makes a real difference, either through local exhaust hoods or enclosed systems. I’ve felt headaches fade once a warehouse fixed its broken fans; proper airflow means less inhaled vapor, plain and simple.
Give acrylic acid space—store it in cool, dry, well-ventilated spots far from heat and incompatible chemicals like strong oxidizers. An old storage shed, uninsulated and out in the sun, nearly caused trouble when temperatures soared. More modern setups use temperature controls and alarms to prevent runaway reactions. Containers should stay sealed when not in use, and staff should get trained to identify leaks or label problems before they escalate.
There’s no substitute for regular drills. Practice keeps panic at bay. Eyewash stations and safety showers should always remain in working order and within easy reach. More than once, someone’s quick sprint to an eyewash station made the difference between recovery and a lasting injury. Companies with a culture of safety—routine checks, up-to-date first aid supplies, fresh training—see fewer incidents and recover faster if anything does go wrong.
Technology keeps pushing solutions forward—better leak sensors, smarter ventilation, improved PPE. Management’s focus on safety makes a difference, but so does worker involvement. Sharing stories, lessons, and near-misses on the workplace floor spreads the knowledge farther than any poster or memo. The payoff is simple: fewer accidents, healthier employees, and less regret when the day is done.
Acrylic acid always pops up in discussions about modern manufacturing. Curious to discover why so many folks get serious around it? Just look at what it does. This small, colorless liquid runs right through the supply chains behind plastics, adhesives, coatings, and even hygiene products. For something so hardworking, it hides a dark side: the stuff irritates eyes and skin, carries a risk of fire, and creates hazardous fumes once temperatures edge up or air gets in.
Many warehouse workers, drivers, and neighbors living around industrial parks understand the risks, even if they haven’t read a safety data sheet. I’ve seen firsthand how accidents ripple: one poorly sealed drum means headaches, missed work, clean-up frenzy, and the kind of stress that lingers. Lose focus, cut corners—suddenly, environmental and human costs stack up in ways that echo for months or longer. There’s no magic to keeping it safe, but honest habits and a little preparation go a long way.
Back in my early days at a logistics terminal, I learned quickly that storing acrylic acid isn’t just about finding empty space and rolling in a barrel. Steel drums and tanks with air-tight seals bring peace of mind but only if they’re kept in check. I met a guy who could spot a weak drum seal by smell alone—he was right every time. A simple missed check once led to a spill; instead of an ordinary Friday afternoon, the air inside the facility burned the back of your throat, and nobody wanted to go home smelling like sour plastic.
Keeping tanks cool and dry stops the acid from getting cranky. A closed system makes sure oxygen doesn’t sneak in because oxidation triggers runaway reactions and nasty vapors. There’s no room for lazy habits. Grounding the storage tanks beats back static electricity that otherwise could spark a fire. Some places go for nitrogen blanketing, forcing an inert gas layer above the liquid. It’s a detail that’s easy to overlook, but if you ask any old-timer, they’ll tell you accidents come from what folks “thought wouldn’t matter.”
Sending acrylic acid on its journey looks simple if you haven’t watched a spill on the highway. Certified tank trucks, rail cars, and containers built for corrosive materials do their job only if every valve and seal has been inspected and re-inspected. Small leaks sometimes go unnoticed at a shipper’s dock but let out fumes or short out gear hundreds of miles down the line. I still remember a colleague who pulled over due to a strange smell—his action kept a minor problem from turning into a roadside disaster.
Well-marked packages never replace clear communication and paperwork that spells out hazards. Trained drivers, prepared emergency kits, and a GPS tracker for each load sound basic, but they make all the difference. Even in the days before digital readouts, a logbook and a commitment to pause if something felt off helped teams avoid bigger trouble.
As tough as acrylic acid is to wrangle, stronger standards and investment in updated tanks, early-detection leak sensors, and ongoing training fix more problems than high fences or thick manuals. Open lines between management and workers allow trouble signs to get flagged early, and bringing first responders into drills strengthens not just companies but whole communities. This chemical won’t disappear from the market; it pays to handle it in ways that put safety, not just schedules, at the top of the list.
Acrylic acid goes by the chemical formula C3H4O2 or, written another way, CH2=CHCOOH. Those numbers and letters stand for a clear, slightly pungent liquid that shows up in a surprising number of parts of modern life. People who work with paints, plastics, adhesives, or even diapers interact with its chemistry in some way. It’s not a glamorous compound, but it carries serious weight in the world of manufacturing.
Acrylic acid strikes with its mix of simplicity and reactivity. One end of the molecule holds a double bond, and the other end holds a carboxylic acid group. That double bond is thirsty for reactions. It grabs onto other molecules and strings them together into long chains, creating polymers. Factories rely on this property—the way it knits together molecules to form plastics and superabsorbent materials. That’s how acrylic acid makes it into products as ordinary as the absorbent crystals in a baby’s diaper or the sticky backing on tape.
This compound has an acrid, eye-watering smell that hardly whispers “everyday product.” With a melting point around 14°C and a boiling point of 141°C, acrylic acid stays usable in most industrial environments without fancy temperature control. It dissolves easily in water, and even low exposures sting the eyes and nose. This explains a lot about how acrylic acid gets handled—factories employ careful storage, steady airflow, and sealed systems to keep it from harming workers. Exposure in high concentrations causes nasty burns, both to skin and to the respiratory tract.
Beyond the nose and skin, the real interest with acrylic acid comes from its ability to jump into chain-making reactions. It’s crazily reactive with bases, alcohols, and other double bonds, turning into compounds that show up under your feet as floor coverings, under your arms as antiperspirant, or in your garden as water-retaining crystals. The molecule sticks together so strongly that its polymers resist breakdown, giving plastics made from it a tough, long-lasting quality.
I’ve toured plastic factories and visited farms where workers spread superabsorbent polymers to help soil hold water. In both places, safety lines every conversation about acrylic acid—people don heavy gloves, seal goggles, and make clear plans for every spill. They put in the effort because acrylic acid produces polymers with high absorbency, clarity, and flexibility. No one’s found a straightforward substitute that delivers on all three fronts, especially at a cost that lets daily essentials remain affordable.
Putting safety front and center matters—nothing about acrylic acid’s physical effects should get brushed aside. Routine monitoring and smarter containment keep people safer, and research continues to chase less hazardous alternatives. Some new labs look to biobased routes or greener solvents, but big shifts need industry buy-in and sharper technology. Until then, acrylic acid’s tough balance of benefits and risks keeps it circling through supply chains and home life, mostly out of sight, but always close at hand.
Factories churn out acrylic acid for everything from paints and adhesives to diapers and detergents. Relying on this chemical keeps consumer goods affordable, strong, and durable. Yet, that same basic substance remains controversial among health professionals and environmental watchdogs. Over a decade in the chemical industry gave me an up-close look at its impact on plants, people, and the world outside factory walls.
Anyone who's spent time around acrylic acid in an industrial setting remembers its sharp, harsh odor and the way even brief exposure irritated eyes and lungs. Short-term exposure—through inhaling vapors or getting liquid on the skin—often leads to burning, watery eyes, a sore throat, and headaches. Those working without proper gear sometimes develop chemical burns or blistering. I watched a coworker ignore safety goggles once and learned the hard way after severe eye pain sent him to the clinic.
There's ongoing debate about acrylic acid’s long-term effects. So far, most studies focus on acute cases. The U.S. National Institute for Occupational Safety and Health classifies it as an irritant. They haven’t linked it directly to cancer in humans, but workers exposed for years still report chronic respiratory issues. If inhalation and skin contact keep repeating, sensitivity and allergic reactions can grow worse.
Factories enforce rules—ventilation, gloves, eye protection—not only to keep people safe, but to minimize lawsuits and loss of skilled labor. Out in smaller workshops and developing regions, these safety steps get skipped far more often. That’s where cases of burns or poisoning turn up more frequently.
What happens inside the building doesn’t always stay inside. Acrylic acid doesn't just vanish; it drifts through air vents and wastewater pipes when not captured or treated. Plants close to release sites sometimes show scorched leaves or stunted growth. In rivers and streams, a big spill can kill fish and foul water for miles. I’ve seen complaints from downstream farmers livid about ruined crops after accidental releases.
Acrylic acid breaks down fairly quickly in air and water, though. That helps limit its reach as long as spills and emissions stay small. Once it seeps into the ground, things get trickier. Low doses don’t seem to build up in plants or fish, but a high concentration can still cause problems for years—especially in places with shallow water tables or heavy rain.
Global demand for acrylic acid shows no signs of slowing. But improved technology offers hope. Closed-loop manufacturing traps and recycles more vapors. Wastewater treatment systems now catch most chemical run-off before it hits the open environment. Smart companies invest in sensors and safety upgrades—automated alarms detect leaks before they become health emergencies.
Stronger rules, steady inspections, and community reporting help keep companies accountable. People who work with the chemical, or live near processing plants, push for transparency about spills and exposures. Some advocates call for developing less-toxic alternatives or using plant-based raw materials, aiming to cut reliance on fossil sources and reduce accidental releases altogether.
Acrylic acid isn’t vanishing any time soon, but awareness, good science, and activism keep the most serious hazards in check. Knowledge, worker training, tight controls, and honest reporting make the biggest difference.