Barium sulfate has a story that takes us back to the days of mineral discovery, when folks first mined barite and tried to wrap their minds around its value. Early on, miners dug up barium minerals without knowing the real potential stashed inside. Over time, chemists figured out just what they had on their hands—by the nineteenth century, barium sulfate started moving into more than just mining records. Artisans noticed it made a decent white pigment for paint. Doctors realized this white powder had more to offer than filling up paint cans, sending it down the path toward medical imaging during a time when X-rays looked more art than science. Progress has stemmed from curious people stumbling across unexpected uses, driven by need as much as discovery. Barite mines fueled growth during periods of industrial expansion, and barium sulfate never really left the scene, instead finding new jobs each decade from construction to medicine.
Barium sulfate may look like just another white powder at first glance, but its uses ripple through technology, healthcare, and manufacturing. Unlike other minerals, it doesn’t just blend in; instead, it finds roles as a contrast agent in medicine, a pigment in paint, a filler in plastics and rubbers, and even as a weighting agent in oil and gas drilling. Hard to dissolve and easy to handle, its appeal stretches across industries. From hospitals to oilfields, this compound keeps popping up in daily life.
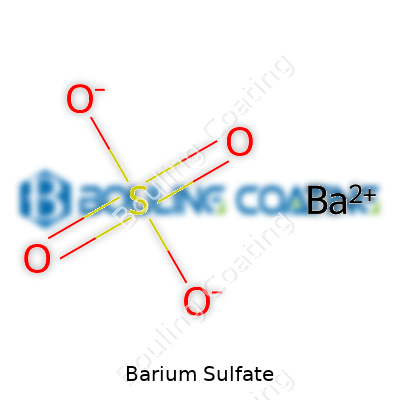
Dense and heavy, barium sulfate carries a molecular weight over four hundred, packing bulk into every grain. This quality comes from the closely-knit lattice of barium and sulfate ions, sticking together in a way that resists breaking up in water or acids. Rain won’t wash it away, stomach acids won’t melt it, and high temperatures just leave it sitting there, looking unchanged. In my hands-on lab experience, barium sulfate always stood out as stubborn and reliable—a white, odorless powder that won’t stain or react much with everyday chemicals. This unusual stability, combined with its bright white color, drives demand for the powder in paints and papers, where staying bright and resisting change is key.
Meeting high standards matters more than ever, especially for a product touching arms of the healthcare industry. Regulations demand tight control over purity and particle size. Medical use calls for powders that don’t wander far above 99 percent purity, with grain sizes measured in micrometers. Dust control becomes a safety point on any site using the powder. Proper labeling draws clear lines about intended use—medical, technical, or industrial—so nobody confuses one grade for another, lowering the risk of accidents. Good labeling, clear packaging, and reliable certification help keep trust with customers who depend on stable, safe ingredients.
Manufacturers lean heavily on mineral barite as the main source, relying on old-fashioned mining and modern purification. Crushing and milling grind down ore, followed by a series of washes to sift off impurities. The real magic happens when chemists set up a double decomposition reaction, letting barium chloride meet sodium sulfate and settle out pure barium sulfate. In industry, controlling reaction temperature, timing, and chemical flow decides what grade you get: super-fine for medical imaging, or coarser stuff for drilling muds. My experience in pilot plants showed just how much tweaking and testing lies behind a finished, consistent powder.
Few compounds play hard-to-get in chemical labs like barium sulfate. It shrugs off most acids and bases. Only super-strong acids, like concentrated sulfuric acid, persuade it to break apart. This stubbornness limits what changes can be made after it’s formed, but that’s also why it stays pure and doesn’t break down in harsh conditions. Engineers have tried surface modifications, like coating powders with polymers, for better dispersibility or specific industrial uses. Blending with pigments can stress the chemical bonds, but barium sulfate usually stands firm—acting more like a tough backbone than a reactive additive.
Barium sulfate knows many names, though most folks just call it by its chemical label. Take a walk through a hospital and you’ll hear it called a “contrast medium” for X-rays or a “radio-opaque agent.” In mines and mineral markets, it goes by “barite,” while in paints and plastics packaging it slips under names like “blanc fixe.” Each name reveals the purpose more than a secret identity—highlighting just how broad its resume really looks.
Handling barium sulfate safely depends more on dust control than chemical risk. It won’t dissolve and slide into the bloodstream like soluble barium salts, but nobody likes a cloud of fine powder in the air. National standards set limits on workplace exposure, and serious manufacturers invest in dust collection and proper respiratory protection. Medical-grade materials face strict controls to lower contamination risks and prevent dosing errors. Experience shows the real dangers come from what might hitch a ride as an impurity rather than the barium sulfate itself. Modern production lines employ particle sizing, rigorous filtration, and spot checks for contaminants like soluble barium, lead, or trace metals.
Anyone who’s had a barium meal or swallow test at the hospital has encountered this compound’s most important medical purpose. Doctors use it for imaging the digestive tract, taking advantage of its opacity to X-rays. Outside of medicine, oil and gas workers rely on barium sulfate as a weighting agent to keep drill bits under control and stop oil wells from blowing out. Paint makers lean on its brightness and inertness to stretch costlier pigments and keep colors lasting longer. In plastics and rubbers, it bulks up the final product without changing essential properties. I’ve seen industrial sites where barium sulfate keeps conveyor belts humming, and hospital radiology wards where it gives answers that more expensive scans cannot.
Today’s researchers look for ways to boost performance in paints, plastics, and new composites. Some try surface modification to enhance mixing in polymers, hoping for lighter, stronger materials. Others tune particle sizes with advanced milling or precipitation, aiming for more vivid paints or clearer imaging. Biomedical engineers dig into better ways to deliver contrast agents, chasing smaller, purer particles to cut side effects or open up new sites for imaging. In both industry and clinics, the push keeps going to refine the old and uncover new uses. Increasingly, research also targets cleaner extraction methods and better recycling, as sustainability pressures build.
Most folks run into barium sulfate through medical imaging, where doctors use it safely. It’s different from other barium compounds, which can cause trouble if swallowed or inhaled. What gives barium sulfate its medical edge is how little of it dissolves inside the body, passing through with minimal fuss. Still, labs track potential impurities, making sure no soluble barium or heavy metals sneak through in finished products. Animal studies and clinical trials keep watch for rare allergic reactions or unexpected side effects. Regulators want rock-solid proof that any material touching the inside of a patient’s gut sits at the far end of the safety spectrum.
Demand keeps growing, driven by the world’s need for energy, infrastructure, and advanced healthcare. Paint and plastic makers see it as a reliable workhorse for cutting costs and stretching the life of their products. Medical researchers trust it for safe, clear imaging, while materials scientists continue poking and prodding for new features or greener sourcing. The biggest challenges ahead focus on improving sustainability, reducing mining impacts, and finding novel ways to recycle or synthesize the compound without trashing the environment. In my own work, I’ve watched as companies shift toward cleaner production, tighter safety margins, and smarter innovation. Barium sulfate’s story isn’t finished—instead, it keeps branching out as technology and industry change.
Walk into any radiology department and odds are you’ll find a bottle of barium sulfate tucked away, often a bit dusty but always ready for the next patient. Hospitals count on this heavy white powder for a simple reason—it’s safe for the body and shows up strongly on X-rays. Doctors who scan the digestive system love it because patients can drink it or swallow tablets without worrying about stomach troubles. From my own time prepping for an X-ray procedure, the memory of that chalky barium shake explains why some folks wrinkle their noses at the mention. Still, for many patients, it’s a small hurdle for the benefits it brings.
Barium sulfate’s greatest strength lies in how it helps doctors diagnose internal problems. The human gut twists and turns in a way that regular X-rays just can't capture well. Barium sulfate blocks the rays, outlining the stomach and intestines like a bold marker. Tumors, ulcers, or blockages suddenly stand out on the results. People often forget the number of lives saved by catching an issue early through simple screening. Almost no other compound delivers such clear contrast without leaving traces that the body struggles to filter out. For those with chronic gut issues, routine scans ease a lot of anxiety, letting doctors see if disease is growing or healing.
Outside of medicine, barium sulfate finds its way into paints, plastics, and rubber goods. I remember walking through a plastics factory once and noticing bags labeled “barite”—that’s the mineral form. It takes a surprisingly small amount to make paint look bright and hide what’s underneath. Contractors won’t settle for a thin look when painting schools and hospitals; a dash of barium sulfate makes sure the old wall never peeks through the new coat.
Companies in oil and gas rely on it, too. In drilling muds, its high weight helps keep oil wells from blowing out, pinning pressure down where it belongs. Despite being bland at first glance, this mineral quietly underpins safety and reliability on many worksites.
Not everything is perfect. Mining barite, the main source, can leave deep scars on the land. Dust from handling the powder also brings risks to workers, though not the same as the more famous barium salts, which are toxic. Facilities that value real safety keep tight controls and provide gear that prevents accidental inhalation. I’ve seen the difference good dust control makes, especially in older factories that took worker safety to heart—lung problems drop off significantly.
Long-term, barium sulfate will likely stick around as long as it keeps outperforming alternatives. Scientists work hard on eco-friendlier replacements for industrial use, but the simplicity and safety of barium sulfate make it a tough act to follow, especially in medical settings. Efforts could shift more toward responsible mining and tighter recycling, balancing continued use with environmental care. Until then, this white powder remains a steady, if quiet, helper in both clinics and factories.
For decades, doctors have leaned on barium sulfate when they need to check out a person's digestive tract. This white powder, mixed with water to make a milkshake or prepared for other routes, shows up well on X-rays. You drink it—or sometimes have it introduced by enema—and suddenly, the path from your mouth to your stomach and intestines lights up for the radiologist. The question for a lot of people: does this stuff belong in your body?
Barium sulfate doesn’t behave like a typical chemical. In normal conditions, your body barely interacts with it. This compound doesn’t dissolve in water or in your digestive fluids, so it usually travels straight through without sticking around or entering your bloodstream. A lot of us would never know what barium sulfate is outside a hospital, but it has stuck around in medicine for one simple reason: it’s reliable and safe for almost everyone when given by mouth or rectally.
Plenty of research backs up that point. Most people can take barium sulfate and walk out after their X-ray, no worse for the wear. Allergic reactions are rare. Poisoning from barium sulfate doesn’t happen in a typical setting, since your gut can’t absorb it. The U.S. Food and Drug Administration lists it as a prescription contrast agent, and imaging centers use strict guidelines when they mix, measure, and deliver each dose. Even for children and infants, trained staff select special formulations and amounts.
I’ve seen family members worry about the process, especially the odd taste and texture. The chalky feel puts off more people than any real physical symptoms. Occasionally, someone will complain of constipation after a barium study, especially if they don’t drink enough water afterward. For patients with certain health problems—like a risk for intestinal tear or a blocked gut—doctors go with other options or skip the test. There’s always a push to make sure people with swallowing trouble or other risks don’t choke or accidentally inhale the mixture, since that can cause serious breathing issues.
Patients deserve to be in the loop with informed consent. A real discussion covers the rare side effects and looks for safer alternatives in folks with special risks. Hospitals have protocols to catch warnings, like anyone who might be allergic to additives in flavored or sweetened forms. In rare cases when the lining of the gut has holes or tears, barium sulfate must be avoided, since it can lead to complications if it leaks out where it shouldn’t go.
As healthcare moves toward more patient-focused care, it’s important to bring these conversations into the room before a test gets scheduled. Patients want to know what’s heading into their bodies, how it works, and what to expect the next day. Nurses are trained to flag people at higher risk of complications, and techs often walk you through the steps before the procedure starts. Most patients, given a clear picture of the process and proper support during and after, manage just fine.
The story with barium sulfate isn’t really about the mystery of a chemical, but about trusting the professional expertise behind its use—and not being afraid to ask questions. For almost everyone who needs an X-ray of the gut, it stands out as a safe tool that helps solve bigger medical puzzles.
Barium sulfate always grabs my attention for its sheer weight and that dense, ghostly white appearance. Most people don’t hold a chunk of this stuff outside of a lab, but if you’ve ever needed a medical X-ray, you probably swallowed a barium meal or had a barium contrast injected. It’s that heavy, white powder that doctors use to see your insides. What stands out is how completely insoluble it is in water. Drop it in a glass, stir for an hour, and you still have the same powder at the bottom. This total stubbornness explains why doctors trust it around the stomach and intestines — it won’t dissolve and poison you with barium ions, which can cause real trouble in soluble form.
We’re not playing with a soft powder here. Barium sulfate rates at about 4.5 on the Mohs hardness scale, so nobody’s scratching it with their fingernails. Its density hovers near 4.5 grams per cubic centimeter — heftier than your average mineral. The crystals take on a tabular or prismatic shape, but in the real world, it comes as a silky, chalky powder. Because it barely reacts with anything outside of molten alkali, it sticks around, handling high heat without breaking down, holding up to most common acids.
That near-zero solubility stems from the strength of the bond between barium and sulfate ions. If you mix it with hydrochloric acid, nitric acid, or water, nothing much happens. For factories and painters, this makes life easy: exposed to rain, sunlight, or bodily fluids, barium sulfate stays intact. Only concentrated sulfuric acid can break it down, and that’s not a spill anyone wants.
I see it most often in radiology departments but that’s just the tip of the iceberg. Some readers may have never thought about where the smooth, bright finish on their car came from. Manufacturers use barium sulfate as a filler in paint, plastics, and rubber to add bulk and improve the gloss. It doesn’t react, fade, or clump, so these materials wind up looking fresher, longer. The dense powder adds heft to plastics—making them less flimsy. I’ve seen this help boost quality in daily items, from floor tiles to brake pads.
Barium sulfate’s unreactive nature also spells safety for workers and consumers. In paints used inside homes or coating pipes that deal with drinking water, the chemical stays harmless. Environmental groups take note of leaching and reactivity, but with barium sulfate, those concerns hit a wall since the molecule won’t separate or seep out in any meaningful way.
Factory workers mention that this powder dusts up easily and can irritate the lungs. It never hurts for companies to invest in better dust management on site, with improved ventilation and enclosed mixing systems. For recycling, engineers could focus on reusing barium sulfate from paint scraps and spent catalysts. Barium isn’t a renewable resource, so recapture and responsible disposal matter.
At the end of the day, barium sulfate holds an odd spot in industry: totally inert, secretly essential, and practically invisible. Its promise comes from what it doesn’t do—no dissolving, no reacting, just staying put whether in our bodies, our paints, or hidden in the mechanics of everyday goods. If you’re looking for a silent collaborator that keeps things running safely and smoothly, there are few elements that can match up to what barium sulfate brings to the table.
Barium sulfate comes up a lot in conversations about medical imaging. People usually talk about the benefits: how it helps doctors see inside the digestive system during X-rays or CT scans. From personal experience and stories exchanged in waiting rooms, what often doesn’t get as much attention are the possible side effects and risks that tag along with it.
The main selling point is that this compound doesn’t break down inside the body. It helps radiologists get a crystal-clear look at things like the esophagus, stomach, or colon. Doctors and nurses say swallowing or receiving barium through an enema doesn’t lead to harmful absorption, and the chemical structure supports this reassurance. Most folks just pass it out unchanged. That said, looking past its benefits teaches us that no medical procedure is flawless.
Most people—myself included—who’ve had barium imaging talk about mild problems that hit after the procedure. Constipation pops up more than any other complaint. That thick, chalky liquid can slow things down for a bit. Drinking extra water helps, but this simple fix isn’t possible for everyone. If someone already struggles with sluggish bowels, things can get tricky. Some people get bloated, others have stomach cramps, and there’s talk of white stools for a couple of days. These little hassles rarely turn into anything worse, but for elderly patients or those with gut issues, even minor complications can become serious.
Complications don’t stop at mild discomfort. Rarely, people breathe in barium during the procedure, leading to a problem called aspiration. This risk grows if a patient has trouble swallowing, such as after a stroke. Lungs don’t appreciate foreign substances. Aspiration pneumonia can follow, and that’s a challenge both for patients and for the medical team handling recovery.
Bowel perforation stands out as a significant risk. If someone already has a hole in their digestive tract from disease or injury, barium should not be used—leakage into the abdomen can cause nasty infections. A strong routine of careful screening before a scan can prevent these events, but mistakes do happen.
Allergy to barium sulfate sticks out as truly uncommon, since barium sulfate itself doesn’t dissolve or react in the body. Still, additives mixed into the formula sometimes cause mild skin rashes or itchy feelings. Health staff stick close for a reason during these procedures; they’re trained to spot dangerous signs like facial swelling or sudden trouble breathing. Every allergic reaction I’ve seen or heard about was caught and treated quickly, but people need reassurance and information going in—not after something goes wrong.
No test or treatment is free from risk, and barium sulfate is no exception. Hydration, open conversations about bowel health, and honest reporting of swallowing problems go a long way. Giving patients time and space to ask questions lifts some anxiety. Doctors can’t promise a zero-risk procedure, and nobody should pretend otherwise. Weighing the benefits and concerns together brings out the best decisions in healthcare, and it helps patients feel respected and heard. That’s what I wish I’d known before my first “chalk shake”—and it’s worth passing on now.
Barium sulfate gets plenty of use, especially in places like radiology departments and labs. Despite its widespread benefits, slipping up on how we store or handle it can bring trouble. As someone who’s worked in environments where safety sometimes falls behind the rush to get things done, I see a lot of value in slowing down and taking a closer look at how we deal with even the most common powders and chemicals.
Trained professionals trust barium sulfate because it won’t dissolve in water or acids and isn’t likely to blow up. Still, letting down your guard with powders rarely turns out well. I’ve watched lab techs breathe in dust just because someone knocked over a container. Barium sulfate doesn’t easily slip straight into the bloodstream, but getting a lungful won’t do anyone any favors. Precaution isn’t about paranoia; it's about keeping yourself and others in good shape.
Crowded storerooms invite corners to get cut. Barium sulfate goes best in a cool, dry, well-ventilated area where moisture can’t make it clump or break down packaging. Good containers, tightly sealed, stop dust from floating around and keep the material from picking up stuff you don’t want in your mix—nobody wants mold or unexpected reactions. Stacking heavy sacks on high shelves usually ends badly, so finding sturdy spots at reachable heights matters just as much as reading a label.
Gloves and basic dust masks aren’t a fashion statement. I remember clearing out an old storeroom where bags had ripped open and nobody thought twice about grabbing handfuls bare-handed. Skin doesn’t absorb this powder the way it does solvents, but irritation and contamination invite unnecessary risk. Goggle up if the dust could go airborne. Spilled powder on the floor asks for a slip or for someone to track it out beyond the workspace, usually right up until someone notices a white smear trailing down a hallway.
Safety gets real once it’s personalized. I’ve seen what happens when care slips—accidents, unexpected downtime for cleaning, and more paperwork than anyone wants to file. These interruptions slow down work, cost money, and shake up trust among teams. Training often fades from memory until routines rework themselves around reality instead of old notes from orientation. Management that gets involved, who show how to use the right tools and organize safe habits, change the culture one detail at a time.
Resources make all the difference. Good signage, accessible personal protective equipment, and regular inventory checks combine to form habits that protect everyone. Communication bridges the gap between different experience levels; that means questions get answered before problems pile up. Local regulations support safer workplaces, but real improvements depend on watchful eyes and people willing to keep learning.
Each container of barium sulfate carries lessons about respect and responsibility. The material won’t cause trouble on its own—trouble enters through inattention or shortcuts. Over time, taking a careful route pays off not only for the tasks at hand but for the people doing them. Safe habits don’t just follow the rules—they build a workplace where everyone goes home as healthy as they arrived.