Butyl acetate’s story stretches back to the roots of synthetic organic chemistry. Chemists started making esters like butyl acetate in the 19th century, using them to mimic fruits’ sweet aromas for perfumes and flavorings. The compound became a staple in the chemical industry during the rise of modern solvents and lacquers in the early twentieth century, standing out for its pleasant scent and solvent power. Paint shops and textile factories helped drive demand, and by the middle of the last century, butyl acetate was a name known in industrial corridors everywhere.
Butyl acetate finds its way into so many products—paints, coatings, inks, adhesives, and even nail polish. Walk through a home improvement store or a nail salon, and you’ll likely smell its distinctive fruity aroma. It thins paints and dissolves resins, working best where artists, carpenters, and industrial painters want quick-drying surfaces and vibrant color. Manufacturers trust it because it leaves behind little residue, speeds up drying, and helps coatings glide on smoothly.
Butyl acetate looks simple on paper: its clear, colorless liquid form makes it look harmless, almost like water at first glance. Its low viscosity lets it flow easily, but its boiling point sits much higher than water’s, making it more persistent in open air. That sweet scent hides a degree of flammability, and workers learn fast to keep it away from sparks and heat. Solubility surprises some: it mixes well with many organic liquids, but only a small amount will blend into water. In everyday terms, it’s a lightweight workhorse with a splash of risk.
In the lab, pure butyl acetate must meet tight standards. Quality checks focus on purity levels, moisture content, and possible contaminants. A bottle destined for an industrial paint facility will get analyzed with gas chromatography, and labeling follows strict rules to make sure nobody gets caught off guard about its hazards. In my own research days, every chemical bottle with butyl acetate wore warning symbols and flammability labels, a constant reminder of the respect this substance demands.
Butyl acetate enters the world through an esterification reaction, with acetic acid and butanol as core ingredients. The process leans on acid catalysts and precise temperature control. Factories scale up by recovering and reusing solvents where possible, pushing for efficiency. Some operations even grab butanol from bio-based sources, aiming for greener chemistry. Streamlining this reaction and minimizing waste matter a lot in the chemical economy—both to cut costs and shrink the environmental footprint.
Chemists love esters like butyl acetate for their reactivity. Under the right conditions, this molecule can break apart or transform, joining the family of derivatives that fill specialized uses. Hydrolysis splits it down to butanol and acetic acid. In more advanced labs, people push butyl acetate into other reactions, sometimes chasing new solvents or flavor compounds. This adaptability keeps it relevant, even as new solvents and regulations change the landscape.
Butyl acetate carries a few aliases: n-butyl acetate stands as its formal name, with IUPAC rules favoring simplicity. Labels sometimes mention “butyl ethanoate” or “acetic acid butyl ester,” especially across different regions. Chemical supply houses list synonyms so buyers know what they’re getting—even if they’re halfway across the world.
Sitting in a busy lab or paint shop, safety takes center stage. Spill a bottle of butyl acetate near open flame, and you’re in trouble. Handling it calls for eye protection, gloves, and good ventilation. Workplaces set strict storage rules, often parking drums of butyl acetate in dedicated storage away from sources of ignition. In high-volume settings, vapor monitoring and fire suppression systems cut risks further. Workplace standards—driven by government rules—set exposure limits and call for regular training.
Painting and coating still keeps butyl acetate in demand, especially for quick-drying lacquers and touch-up sprays. Printers rely on it to keep inks from clogging heads and to help colors shine on magazine covers. Labs occasionally use it as an extraction solvent, picking up nonpolar compounds for analysis or purification. You’ll even find its distinctive fruit notes in flavor and fragrance work, though always in controlled, tiny amounts. I remember using it in the lab for thin-layer chromatography—its solvency power shined when separating tough samples.
Scientists tinker with new blends to create safer, more environmentally friendly coatings, and butyl acetate still plays a big role. The push for water-based paints means some places seek substitutes with vapor profiles less hazardous to workers, but the balance between performance, safety, and price remains tricky. Some research teams experiment with bio-based butanol to make greener butyl acetate, hoping renewable sources will keep it relevant in tightening regulatory environments. Others work on recovery and recycling instead of letting fumes go unused or wasted.
Butyl acetate rarely makes headlines for toxicity, but neglect leads to trouble. Inhalation of concentrated vapors brings headaches and dizziness. Prolonged exposure makes skin and eyes burn, so long-term safety means more than just gloves and goggles—it means proper ventilation and careful handling every time. Animal studies show low long-term toxicity, although research always digs deeper, hunting for subtle effects workers might miss in a fast-paced plant. Regulators and occupational safety experts draw the line at levels meant to keep both acute and chronic effects low. Through my own experience teaching lab safety, stories about careless handling often stick far more than dry numbers on a chart.
Butyl acetate won’t fade away soon. Many industries lean on it because it gets the job done where others can’t. Fears about air quality and chemical exposure drive new paint and ink technology, but replacing butyl acetate with safer, renewable alternatives remains a work in progress. Efficiency gains might help, using technology that captures or reuses vapors instead of losing them to the air. As the green chemistry movement grows, research teams push boundaries—seeking new ways to make old reactions cleaner, faster, and safer. Watching this progress unfold, I see a future where butyl acetate’s legacy shapes how chemists balance reliability, safety, and a lighter footprint on the world.
Most folks don’t recognize the name butyl acetate unless they’ve spent time in a paint shop or chemical plant. Yet, this colorless liquid shapes much of what we see around us, especially if you like art, cars, or a fresh coat of something new. I realized its reach when I started painting furniture as a side project. The scent that hung in the air—the one that lingers for days—comes from butyl acetate doing its job.
Take a look at a newly painted wall, a car rolling out of the shop, or even a shiny metal cabinet. Butyl acetate often sits in the can, helping drift those pigments and binders into a smooth, even finish on surfaces. It dissolves stubborn resins and dries at just the right pace, which brushes streak out and spray guns distribute evenly. I found that using paints with this solvent makes cleanup and application far less frustrating, especially in humid weather.
Walk into a nail salon and the sharp scent greets you before you spot the polish racks. Nail polishes, removers, and some cleaners rely on butyl acetate to thin out thick formulas and keep things glossy. Perfume makers lean on its almost sweet smell to help carry fragrances without overwhelming the nose. It still gets into adhesives, inks, and even flavoring workrooms, though only folks on the production lines catch that detail.
Spending a few afternoons in a poorly ventilated studio taught me one lesson—solvents aren’t just innocent bystanders. Prolonged breathing of butyl acetate fumes leads to headaches, dizziness, and short-term memory issues. Some days, I opened the window a crack and wore a cheap mask, but that didn’t cut it. To protect health, workplaces enforce fresh air flow and hand out respirators. Limiting time around open cans keeps problems to a minimum. The chemical fades quickly from finished objects, but those handling it day in and out face more regular exposure.
Butyl acetate makes modern coatings work and helps keep beauty products effective. Yet, the industry pushes toward options that protect both people and the environment. Environmentally friendly paint lines reduce the volume of strong-smelling solvents without skipping durability. Some labs test bio-based versions made from renewable crops, putting less stress on fossil fuels and human health over time.
Using butyl acetate in your own projects deserves a little respect for its power. Find a spot with a breeze, wear gloves, and give your tools time to air out before packing them away. If you work in a place heavy with industrial scents, learn the safety protocols by heart. Industry leaders, researchers, and everyday people keep nudging manufacturers to cut down risk and boost renewable alternatives. Change happens slowly, but staying informed on the chemicals we use gives us a say in pushing for safer choices.
People often don’t realize how close chemicals are to their routines. Butyl acetate shows up in workplaces, garages, and even nail salons. Its name might sound industrial, but its role stretches from making car paints dry smooth to giving fruity odors to flavors. As someone who spent a decade working around auto-body shops and art studios, the sharp, sweet smell of this solvent still sticks in my memory. But even if you don’t notice it, exposure can sneak up through smell, skin, or air.
Research highlights clear risks. Breathing it in can irritate eyes, nose, and throat, sometimes setting off headaches or dizziness. Some workers recall a sickening pressure after painting cars in stuffy shops—a feeling that lifts in the open air. People working without good ventilation run the biggest risk, but home hobbyists spraying paint or using lacquer thinner might not fare much better if the garage door stays shut.
Health authorities like the CDC and EPA recognize that regular exposure, especially in high doses, can harm both the nervous system and lungs. Those with asthma or allergies could have worse symptoms. Direct skin contact also dries or irritates, leading to rashes after spills or careless handling. I still remember how one old coworker, too eager to clean up a spill, developed a nasty red patch across both hands. We all learned to grab gloves after that.
The science on cancer connections or major organ problems from low-level exposure still leaves some gray areas. Toxicology reviews agree: most immediate danger comes from breathing in a lot at once, not from faint traces in everyday situations. Still, no one likes being the test case, and cumulative risk grows in jobs with daily use.
Industry isn’t blind to these dangers. Workplace rules in many countries lay out clear ventilation standards and suggest personal protective gear. OSHA and similar groups mandate exposure limits for a reason, recognizing that fast-evaporating solvents belong in well-aired spaces, not in your bloodstream.
Protecting health starts with basic habits. Good ventilation matters most. A small window fan or open garage door can make all the difference. Gloves and goggles, not oversized for the job, help cut skin contact. Paint shops and factories often use respirators for a reason, and they aren’t just for show on safety posters.
I’ve learned that education beats warning labels. Workers and even home users need real training on handling containers, cleaning up spills, and choosing safer substitutes whenever possible. Some industries shift to less toxic water-based products; even artists look for “green” paints that cut down the fumes. This isn’t just regulation; it’s respecting the bodies that do the work.
People won’t likely toss butyl acetate altogether, but everyone—boss, worker, DIYer—can take steps to limit the hazards. Awareness, smart work habits, and safer options keep health from being the price of a good paint job or a shiny set of nails. Nobody has to walk away dizzy or itching just for doing their job or working on their car. Safety tools exist, but only if people use them.
It's easy to forget just how crucial safe storage becomes in both factories and workshops storing chemicals like butyl acetate. As someone who’s walked through busy paint and coatings production floors, I’ve seen up close what happens when the basics get overlooked: sharp odors, headaches, and fire hazards can show up before you know it. Butyl acetate lands on the shelf in more industries than people realize—paints, nail polish, soaps—so it finds its way into all sorts of workplaces, not just chemical labs.
Butyl acetate is known for its strong fruity smell, which is pleasant enough in nail polish remover, but gets overwhelming fast if storage goes wrong. Forget for a moment about complex regulations and focus on what regular folks need to know: this is a flammable liquid. If someone stacks drums in a hot warehouse or lets vapor collect in a stuffy room, it sets the stage for disaster. Spark from an old light switch, and suddenly that minor shortcut spells big trouble.
Experience shows that the right spot for butyl acetate combines cool, shaded areas and plenty of airflow. A storage room with open windows, industrial fans, or vents gives vapors no place to build up. Moving drums away from forklift traffic—instead of just cramming them against a wall—reduces dents and spills. Sealing containers tightly keeps both fumes and accidental splashes where they belong.
Steel drums do the job, but only with rust-free tight lids. A single cracked seal means more than a mess; leaks can lead to slippery puddles or even long-term environmental harm if chemicals seep into the ground. From my own visits to warehouses, a simple leak tray under drums and cans—rather than stacking them directly on old wooden pallets—makes cleanup easier and keeps any problem from spreading. People who handle the product for a living know a splash can sting skin and make eyes water, so putting butyl acetate in a locked cage or behind a marked barrier isn’t about paranoia, but about giving everyone a fair chance to work safely.
Firefighters who’ve answered calls at industrial parks stress one point: butyl acetate burns fiercely once it gets going. Keeping storage spaces far from heating units, open flames, or places where people smoke cuts down on needless risks. Having fire extinguishers nearby, especially foam or CO2 types, makes sense for anyone moving barrels around. Mixing chemicals together, even accidentally, can create fumes nobody wants to breathe or reactions that send flames racing across the floor. There’s a reason so many safety posters show dramatic images—these things really happen.
Strict rules are written for good reason. Workers need clear labels, not bits of tape and fading marker. Posting a big, visible sign near the entrance keeps new hires—who might not know better—from wandering in with a phone or a lighter in their pocket. Regular training sessions are a must, not just paperwork. Even seasoned staff forget things when they’re under pressure, so managers play their part by running drills and refreshing everyone’s memory.
Storing butyl acetate goes beyond following a list in a manual. It calls for noticing hazards other folks overlooked and fixing small issues before they grow. Simple habits—ventilation, fire safety, clear labeling—go a long way. They protect both people and business, a lesson learned time and again across workplaces large and small.
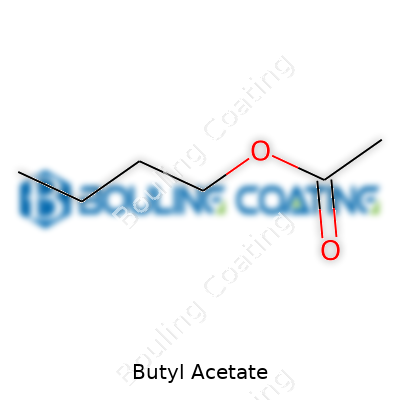
Everybody who’s spent time around paints or nail polish remover has probably caught a whiff of something fruity, sweet, and slightly stinging. That scent often comes from a solvent called butyl acetate. In the lab, it’s written plainly as C6H12O2. What’s refreshing about this formula is how it lays bare the backbone of real-world chemistry: unglamorous but totally essential to how things come together in industry and daily life.
Butyl acetate’s structure roots itself in two building blocks: butanol from the butyl side and acetic acid from the acetate part. This bond gives us a compound that powers everything from fast-drying paint in old houses to cleaning agents in electronics. The formula tells you how many atoms are in a single molecule—six carbons, twelve hydrogens, and two oxygens—but behind those numbers sits a lot of hard work keeping products reliable and consistent.
For students and folks working in labs or on factory floors, formulas like C6H12O2 are more than academic trivia. They’re blueprints for safety, quality control, and troubleshooting. If a company accidentally grabs pentyl acetate thinking it’s butyl acetate, paint dries wrong. The wrong solvent can gum up machinery or cause batches to fail safety checks.
In my own experience working with adhesives, knowing you’re actually handling butyl acetate keeps reactions predictable. Years ago, checking the formula on a mislabeled drum of solvent before mixing it in a batch saved us from a whole week of lost production. Double-checking that C6H12O2 showed up alongside “butyl acetate” on the label made the difference between smooth sailing and a costly setback.
The structure of butyl acetate isn’t just about making things stick together or cleaning up grease. It steps into the conversation around safety and indoor air quality. Occupational limits exist for a reason: breathing too much vapor can hit the nervous system, producing headaches and drowsiness. Understanding what’s in the air and on your hands helps everyone stay healthy, especially in paint shops and nail salons.
Fact sheets and regulatory documents pull that C6H12O2 out every time for clear labeling. It’s an anchor point for hazard training, environmental monitoring, and chemical tracking. When communities talk about safety data sheets, they want transparency. Knowing precisely what’s in a can or bottle helps workers make smarter choices—like swapping in water-based alternatives where possible, or investing in better ventilation.
Basic chemistry, like the makeup of butyl acetate, helps companies, builders, and end users reduce mistakes. Digital inventories log C6H12O2 so warehouse managers don’t mix up containers. Manufacturers can trace contamination to the source if the paperwork ties the incident to a specific batch formula. And for those seeking greener options, formulas offer a clear way to compare alternatives before they hit production lines.
All told, butyl acetate’s chemical formula may look plain, but it carries a surprising amount of weight in real decisions. By knowing the structure, everybody from teachers to technicians gets a firmer grip on what they’re working with and what’s at stake for quality and health. This isn’t about memorizing symbols for the sake of it—it's about keeping workplaces safer, products reliable, and science honest.
Most folks who’ve worked in paint shops or printing rooms have crossed paths with butyl acetate. That sweet, fruity smell gets into your clothes and sticks around long after you leave. At a glance, it doesn’t seem deadly—some might even mistake it for a casual nuisance. That’s why people let their guard down, and trouble follows when small habits slip.
Butyl acetate can do real harm when basic practices go ignored. Breathing in its vapors for too long can leave someone dizzy or headachy. Anyone who’s ever stripped lacquer in a tight garage remembers that spinning feeling if the windows aren’t open. In high amounts, it can even mess with your nervous system.
The stuff burns fast, too, and will catch fire before you hit the boiling point of water. Sparks from cheap power tools or a half-lit cigarette can start something nasty if it’s in the air. Setting aside the risk for a lunch break just means coming back to a building that could have gone up in smoke.
This liquid isn’t gentle on skin or eyes. Splash it once, and you know exactly why gloves aren’t just decoration. Folks who go without find their hands drying, itching, or peeling in ways that linger. Worse if it gets in your eyes—quick flushes help, but the sting can hang around. People scoff at safety glasses until a stray drop lands, then they line up for protection without a word.
A few basic routines work better than any complicated rulebook. Workers should crack open windows and flip those fans on before the job even starts. That sweet smell serves as a warning; if you can taste it, you need more air moving through the space. It helps to label containers, so nobody refills a coffee cup out of the wrong jug. I’ve seen that mix-up in real shops, and the fallout is always messy.
Spills will happen—having some absorbent grit or pads nearby makes clean-up quick, and keeps slippery pools from spreading across the floor. It pays to store butyl acetate in closed, grounded metal cans away from sparks and direct sunlight. You’d be surprised how much a forgotten rag or dirty shirt can fuel a blaze. Tidy lockers aren’t just a nod to neatness—they keep everybody safer.
Nobody learns safe handling out of a manual alone. Clear demonstrations from someone experienced go further than signs on the wall. Sharing stories about close calls—without shame or blame—helps everyone remember the hidden dangers before cutting corners. Shops with open dialogue build trust, and that keeps more people in one piece over the long haul.
Proper ventilation remains key. Upgrading exhaust fans, even if it means a bigger bill up front, turns out cheaper than treating injuries or cleaning up after an accident. Personal protective equipment should be ready at every doorway, not locked away in a manager’s office. Regular team check-ins work well—if someone’s feeling woozy or forgetful, it’s a cue to take a break.
Butyl acetate rewards respect and common sense. Give it a little forethought, build habits from experience, and you claim the benefits without courting disaster. The folks who last in this business know that keeping safe is like sweeping up at closing time—you might not notice the effort, but you sure notice the mess if you skip it.