Calcium carbonate’s story stretches back deep into geological time, far before anyone ever held a flask or ran a chemical assay. It’s a core ingredient in the layout of our natural world—limestone cliffs, marble temples, and even the chalk used in prehistoric cave art. Scientists in ancient civilizations already noticed its presence and usefulness, but real progress started once chemistry entered the scene in Europe. In the 18th century, advances in mineralogy and basic chemistry started revealing the secrets behind common minerals, pinpointing calcium carbonate in everything from seashells to massive stone quarries. The discovery that heating it produced calcium oxide and released carbon dioxide shaped not only chemistry lessons but entire industries. Masons, farmers, and painters saw the value in it long before anyone wrote a chemical formula. Even today, there’s something almost poetic about recognizing the link between the mineral that built the Pyramids and the antacid that calms a sour stomach.
Everyone interacts with calcium carbonate almost daily, even if they don’t know its name. Toothpaste grit, baking powder, cement, dietary supplements, paper gloss, and plastic finishes each owe something to this one mineral. Some folks use the precipitated form for ultra-fine applications in pharmaceuticals and foods, while others turn to ground limestone for construction. Its prevalence flows from how abundant and easy it is to process. The world pulls millions of tons from quarries every year, shaping it for applications that touch agriculture, medicine, art, and industry. Companies keep refining how pure, how white, how granular, and how safe they can make it for more specialized uses. That constant refinement creates jobs, research opportunities, and even an international market rooted in something as simple as ground-up stone.
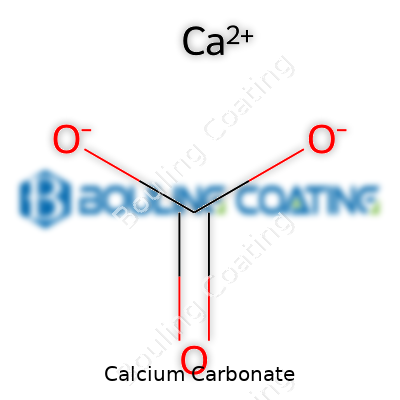
Everyone who’s ever picked up a hunk of chalk or admired a marble sculpture has seen calcium carbonate’s milky white look. It’s tasteless, free-flowing, and stable under standard conditions. Chemists know it as CaCO₃, marrying calcium, carbon, and oxygen into a crystalline structure that’s hard and dense as marble or soft as chalk, depending on how it grew. It barely dissolves in pure water, which makes it sturdy in art and construction, but let a little acid in—like vinegar or stomach acid—and it starts fizzing, breaking down to form carbon dioxide bubbles. Its low solubility makes it a slow-acting soil amendment, but its reliable reaction to acids underpins medical, food, and industrial uses. The stable performance, the reliable whitening effect, and its chemical predictability mean people return again and again to the material in everything from tablets to paint.
Packing a bag of ground limestone or a bottle of calcium supplement comes with a raft of rules these days. Stringent labeling for pharmaceutical, food, and feed-grade products matters, because consumers want to know purity, particle size, heavy metal content, and any added ingredients. Finer grades turn up in pills and toothpastes—the sort that leave no grit behind. Big players in construction and plastics care more about brightness, density, and shape. International standards from USP, FCC, and the EU require detailed test results before a batch can ship, so labs run spectral analyses, weigh trace elements, and confirm there’s no out-of-place trace of lead or arsenic. Even the coatings on tablets, or whiteners in paper, get tracked closely, since regulators have long memories about history’s chemical accidents. Rigorous standards prevent shortcuts and keep consumers safe, especially where health is at stake.
Preparing calcium carbonate for industry sounds simple: dig, crush, screen, sometimes heat. Yet real expertise lies in the details—choosing the right quarry, grinding to the right size, and washing to remove impurities. Plants that make high-purity versions for supplements or science use chemical precipitation, reacting calcium chloride or calcium hydroxide with carbon dioxide under carefully controlled conditions, then filtering, drying, and milling the result. Farmers coax out calcium carbonate by burning limestone in kilns, then hydrating the quicklime to form hydrated lime if needed. Each step—extraction, purification, drying, grinding—draws on both tradition and modern process control. In my own experience working alongside plant operators, the key differences often come down to attention to detail, respect for safety, and understanding which small changes deliver major gains in quality. This hands-on know-how supports everything from safe food to resilient bridges.
The basic chemistry of calcium carbonate revolves around its gentle stubbornness. Dump it in water, and it lingers as a fine suspension, refusing to dissolve. Toss in acid, and the familiar fizz of carbon dioxide appears, a reaction behind both natural cave formation and the fizz in antacids. Heat it at the right temperature, and calcium carbonate gives up carbon dioxide, leaving behind calcium oxide—a building block for mortar and a thirstier, more caustic chemical player. Modifications for plastics and rubber often see the surface coated with stearic acid or silane compounds, making the mineral friendlier to polymers and less likely to clump. The interplay of simple chemistry and clever surface engineering means scientists can turn a common mineral into a high-performance additive, all while producing less dust and waste for manufacturers. These tweaks might seem technical, but even small changes breathe new life into age-old materials.
Anyone reading ingredient lists or technical datasheets knows calcium carbonate wears disguises. The old names like limestone, marble dust, and calcite often signal raw geological sources, while precipitated calcium carbonate (PCC) or ground calcium carbonate (GCC) describe the way the mineral’s prepared more than what it is. Chalk remains a familiar label from education and children’s art. Food-grade batches go by E170 in Europe, bridging language gaps across regulatory regimes. Drugstore antacids often lean on commercial branding, but the core chemical can also hide behind terms like whiting or agricultural lime. Names change for marketing and regulatory needs, but the molecular skeleton stays the same.
Working with calcium carbonate doesn’t mean throwing safety out the window. Dust inhalation can grate the airways, though toxicity sits pretty low compared to many industrial chemicals. Industry guidelines stress dust control, personal protective equipment, and good housekeeping in plants and workshops. Material safety data sheets rule out risks from casual contact but push for good ventilation and regular cleanup. Food and pharmaceutical applications demand zero tolerance for pathogens or heavy metals, putting more weight on batch testing, clean production lines, and regular audits. Keeping silica and asbestos contamination out is just as important as limiting trace metals, since impurities crown the danger list. Effective oversight and investment in equipment mark the line between a safe workplace and health complaints or recalls. In my own experience, companies who invest early in prevention rarely need to scramble after problems catch up to them.
Calcium carbonate found its way into nearly every sector with a material to fill or brighten—paper mills, plastics factories, paint shops, agriculture, pharmaceuticals, and even the sugar industry. In agriculture, spreading pulverized limestone tames acid soils and strengthens crops with calcium nutrition. In medicine, it holds its ground as a gentle antacid, a calcium supplement, and a binder in tablets. The paper and card industries lean hard on it for smooth, sturdy sheets and bright finishes. Rubber and plastics get bulked out and stiffened without much cost. Even in paints, it helps pigments spread and stay bright. One remarkable lesson from seeing these industries up close is that the small mineral flows that start in rural quarries often end up woven into the fabric of daily city life.
R&D labs don’t overlook minerals like calcium carbonate just because their chemistry seems familiar. Today’s research teams dig into how micron-scale tweaks in particle size and shape can alter paint flows or improve tablets. Some labs even engineer the mineral’s surface to anchor vitamins or control drug release, designing composites and coatings that respond to pH changes in the body. Nanostructured forms show promise for drug delivery or pollution control, where a thousand times more surface area gives a massive boost to effectiveness. Paper coatings keep evolving as companies hunt for lighter, brighter finishes. The primary barrier to progress often comes less from the chemistry, and more from cost and reproducibility at scale, yet the most creative researchers keep finding ways to cut energy inputs, recycle byproducts, and boost performance in end uses.
By toxicity standards, calcium carbonate stands out as a fairly benign player compared to a host of industrial materials. Ingesting dietary or pharmaceutical grades proves safe in reasonable doses, explaining its long history in the medicine cabinet. Inhalation of fine dusts still presents an occupational hazard, stressing the need for dust controls in mining and manufacturing. Scientists investigating chronic exposure rarely find alarming trends unless silica, trace heavy metals, or crystalline contaminants ride along with the mineral. Research into environmental impact tends to focus on accidental overuse in soils, where runoff can alter local water chemistry and trigger unwanted blooms of algae. Overall, strict adherence to testing for purity and dust containment means the real dangers come less from calcium carbonate itself and more from neglecting proper handling or using contaminated sources.
Calcium carbonate might seem like a basic raw material, but its future looks open-ended. Lightweight plastics and papers call for cleaner, finer fillers to save weight and energy. Green building pushes for sustainable concrete, where optimizing the chemistry of limestone becomes more urgent. Carbon capture and storage have researchers tinkering with ways to lock CO₂ into durable carbonates, turning climate challenges into construction solutions. Biomedical teams keep exploring nano-calcium carbonate for drug delivery and imaging. Industries under pressure from both cost and climate keep investors hunting for tweaks that squeeze out waste and raise value. All this progress rests on a deep knowledge of mineral chemistry fused with hard-won practical experience, and that’s exactly where the promise of calcium carbonate continues to grow.
I grew up thinking chalk was just for the blackboard. I was wrong. Chalk, or what we know as calcium carbonate, pops up in the most unlikely corners of daily life. Start in the kitchen. Most toothpaste tubes boast about whiter teeth; a look at the ingredients almost always finds calcium carbonate. Dentists trust its light grit for brushing away plaque without ruining enamel. Skipping over to the pantry, you’ll notice antacid tablets. Those tablets you chew when your stomach starts complaining? Same compound, quietly neutralizing acid, letting you go back to regular life.
Farms depend on this mineral too. Many soils turn too acidic for crops to grow well, which threatens entire harvests in some regions. Spreading crushed limestone over fields reverses that acidity. It isn’t about fancy tech; it’s about basics. Crops thrive, food yields rise, and fewer families go hungry. The story doesn’t end outside. Processed cereals and non-dairy milk use calcium carbonate to replace the nutrients missing from grains or artificial alternatives. People who swap cow’s milk or grains for plant-based versions often have no idea where that extra calcium comes from—it’s the same mineral squeezed out of rocks, then scrubbed and sifted before showing up in breakfast.
Walk around your home, look at the walls, and chances are, they contain calcium carbonate. Plaster, cement, and even some paints rely on this mineral. Manufacturers use it to make surfaces smooth, solid, and long-lasting. The approach isn’t new. Ancient Romans mixed limestone dust into their famous buildings. Builders trust it because it’s strong and cheap; people living under these roofs appreciate comfort and security.
Schools around the world use chalkboards every day, but learning doesn’t stay in the classroom. Calcium carbonate plays a subtle role in clean water systems, since it helps remove impurities. In the paper industry, it brightens pages, hides eye-straining blemishes, and keeps the cost down, which matters for cash-strapped schools. The plastic you hold in your hands to carry groceries or shield your phone screen—manufacturers fill it with calcium carbonate to boost strength without raising the price. This mineral quietly makes life smoother, safer, and more affordable.
Sometimes it feels we only notice minerals when they run short. Calcium carbonate offers clear benefits, but digging it up leaves scars on landscapes and pushes up carbon emissions. More recycling in construction and packaging can lower pressure on natural deposits. Finding ways to reuse old building rubble, for example, means less rock pulled from fresh quarries. People often don’t see their connection with the planet beneath their feet, yet choices about the raw materials in everyday objects ripple out to distant places. Companies and consumers should both look past short-term convenience. Smarter choices about sourcing and recycling help communities and cut waste. It isn’t magic, just practical action that won’t break budgets, but can help keep the things we depend on affordable and reliable for years to come.
Calcium carbonate pops up in daily life more often than many folks notice. It shows up in antacids, chalk, chewable calcium supplements, toothpaste, and even in some plant-based milks. If you’ve chewed on Tums for a sour stomach or tried to up your calcium intake with a supplement, you’ve already added this chalky mineral to your diet.
Calcium is no stranger to the human body. Bones and teeth rely on it, muscles need it to move, and nerves use it to send signals. Calcium carbonate provides a way to increase calcium through food or pills. Stomach acid helps break it down before calcium travels to where it can be absorbed. This mineral works best for people who don’t have low stomach acid levels, such as younger folks. Age or certain medications can blunt stomach acid and slow down the process.
Doctors often recommend calcium carbonate for people who don’t get enough calcium from food. Dairy allergies, plant-based diets, or health conditions that rob the bones—these are cases where supplements do help. The FDA recognizes calcium carbonate as safe when used within dietary guidelines, and medical experts rely on it to help those with weak bones or stomach acid issues.
Problems crop up when people go overboard. Taking too much can bring on bloating, gas, constipation, and even kidney stones in folks who already run a little high on calcium. Too much calcium can also block the body from using iron or zinc, so the chances of other nutrient problems can go up.
No matter what’s on the bottle, safety always traces back to the production process. Medicine-grade calcium carbonate goes through purification that garden chalk doesn’t see. So, eating antacids is very different from scraping chalk off a blackboard. Trusted brands and regulated products lower the risks of harmful contaminants like lead or heavy metals.
It’s also worth paying attention to other ingredients. Some pills carry artificial flavors, sweeteners, or colorings. People with allergies or sensitivities should read the label if possible.
I’ve met many who worry about “calcium overdose” after reading about risks online. Anecdotes make the rounds quickly, but big health surveys show that most adults don’t reach the danger level through supplements alone. To avoid overdoing it, mix dietary sources—dairy, leafy greens, nut milks—with smaller supplements if needed. Talking with a doctor clears up confusion, especially for folks managing complex health issues.
Not everyone needs a pill to fill the gap. Small changes, like adding broccoli or almonds to meals, can sidestep the problem altogether. For those on acid-reducing medicines, calcium citrate absorbs more easily. Reading up on food labels and asking questions gives anyone the power to make good choices for their body.
Growing up, my relatives leaned on chewable calcium for bone strength, especially women in the family. Their experience matches what science says: used as directed, calcium carbonate does its job. It feels like another reminder that common sense and a little professional advice often sort out confusion. Taking a closer look—not just swallowing blindly—always pays off in the long run.
Calcium carbonate lines the shelves at every pharmacy. Many people reach for it, wanting stronger bones or hoping for quick relief from heartburn. It’s a mineral that seems safe and familiar, but the story doesn’t end at strong teeth and happier stomachs. The way we use it, the amounts we take, and our own health all play a part in how our bodies react.
Most folks take calcium carbonate without trouble. Stomach troubles crop up more than people expect though. It can bring on constipation, especially when too much piles up in a daily routine. Those tablets block acid in the stomach, which slows digestion and sometimes leads to hard stools. People who rely on it daily—usually older adults or those taking it for bone health—tend to notice this more.
Bloating and gas don’t get talked about enough. A fizzy feeling or extra burping can follow, especially if tablets dissolve too fast in the stomach. This isn’t the worst problem, but it can leave people feeling out of sorts.
Doctors see side effects spike when folks overdo it. Taking more calcium than needed can throw off the body’s delicate balance. Too much calcium can lead to a build-up in the blood, called hypercalcemia. That might start with nausea and vomiting, tiredness, or even confusion. Left unchecked, high calcium levels stress the kidneys. Some people end up with kidney stones, which are as painful as anything.
Hypercalcemia doesn’t show up overnight. People with kidney problems or those taking certain medicines face higher risks. Thiazide diuretics, for instance, can tip the scale, trapping more calcium in the blood. This is why checking in with a doctor if you’re mixing supplements and prescriptions is more than just a formality—it’s a real safety net.
Years back, my grandmother started taking calcium carbonate on her own because her neighbor swore by it for bone strength. She didn’t read labels, just popped what looked like the right number of tablets. It worked for a while, but then she started feeling achy and tired, almost flu-like. Her experience isn’t unique. Many people add over-the-counter supplements, thinking more can only bring more benefit. This thinking runs people into trouble, especially with something as available as calcium carbonate. It reminded all of us that even the most common supplements need respect and a bit of extra attention.
The conversation about calcium carbonate isn’t just about problems. Used wisely, it can serve real health goals—bone density and relief from sour stomach. The trouble starts when people leave their doctors out of the loop. Doses creep up, or they ignore how radically other prescriptions might interact.
Reading labels, talking with health professionals, and keeping a running list of medicines and supplements takes time but pays off. It helps avoid common side effects and steers clear of the worst-case scenarios. Making these changes becomes so much easier after you see someone close struggle.
Supplements don’t fix everything. They work best as part of the bigger picture, one that respects how every body works just a bit differently. Staying informed, honest, and cautious gives you the best shot at better health—without the nasty surprises.
My mother broke her hip last winter. She’s the kind of person who never sat still, always in the garden or fussing over grandkids. Suddenly, her hospital room filled with pamphlets about calcium and osteoporosis. As someone who thought drinking the occasional glass of milk kept bones safe, that accident served as a wake-up call to take calcium more seriously.
No question, our bones need enough calcium to stay strong, and women, especially over 50, run a higher risk of bone loss. Data from the National Institutes of Health show that nearly half of adults in the US don’t get enough calcium from their diets. That shortfall opens the door for supplements like calcium carbonate, widely available and not expensive.
Many people, myself included, used to think you could just pop a calcium pill anytime you remembered it. Turns out, calcium carbonate absorbs best when taken with food. The acid produced while eating helps the body pull more calcium from the supplement. If you only rely on coffee and toast at breakfast, consider pairing your supplement with a proper meal, something with a little protein and healthy fats.
There’s such a thing as too much at once. The body only absorbs a limited amount of calcium at a time — usually about 500 to 600 milligrams. Taking more in one go won’t give you a bigger boost. Instead, split your dose and spread it out through the day. Breaking up the routine this way got easier for me once I left reminders on my phone, rather than depending on my memory.
Some medications don’t mix well with calcium carbonate. Thyroid medicine and some antibiotics, for example, can lose their potency if taken too close together. Double-check with a pharmacist or doctor about drug interactions, especially if you have a complicated medication routine.
Too much calcium from supplements can also lead to problems. Kidney stones, constipation, and even issues with heart health become more likely at higher doses. The Recommended Dietary Allowance for adults usually falls between 1,000 and 1,200 mg per day from all sources. If you already eat lots of dairy or leafy greens, your supplement needs may be smaller.
If you have difficulty swallowing large tablets, chewables or powders mix easily into yogurt or smoothies. That tip made all the difference for my mother. Vitamin D partners well with calcium, helping your body grab more out of each dose. Many supplements now package these together, which saves time and effort.
Reading nutrition labels helps cut through guesswork. Many cereals, plant milks, and orange juices add calcium by design, so keeping a daily tally paints a clearer picture of what you actually need. A quick food diary for a week gave me plenty of insight into where my diet fell short.
Healthy bones take more than just chasing numbers. Weight-bearing exercise, a diet rich in leafy vegetables, and listening when your joints complain all play a role. My experience boils down to this: talk to your healthcare provider, watch your diet, keep a regular schedule, and don’t be shy about asking questions at the pharmacy counter. Good habits, built one small choice at a time, can protect you far longer than any single pill.
Many folks grab calcium carbonate for two big reasons—building bone strength and taming heartburn. It sits on store shelves as chewable tablets or chalky powders. For most people, it looks as harmless as a glass of milk. It surfaces on doctor's lists, pharmacy shelves, and kitchen drawers everywhere.
Plenty of us add supplements like calcium carbonate without a second thought. Over-the-counter means safe, right? Not quite. The body juggles more than food and supplements every day. Mix in daily medications, and things get complicated fast.
The stomach’s acidic environment helps break down pills, but calcium carbonate lowers that acid as it fights heartburn. Some medicines, especially certain antibiotics and antifungals, rely on those acids to dissolve. Take calcium carbonate at the same time, and the medicine may not do its job. A real-world story—one of my relatives learned this lesson while trying to treat a stubborn stomach infection. The antibiotics stopped working properly because of regular antacid use. A switch in timing fixed the problem.
Doctors still see patients lose control over thyroid conditions, blood pressure, or even mood disorders, all because a supplement stepped in at the wrong time. Levothyroxine (used for thyroid problems) binds with calcium and can’t get absorbed as intended. Blood pressure meds called calcium channel blockers might sound compatible but can behave unpredictably if extra calcium enters the system through supplements.
A trip to the pharmacist or primary care provider often ends with a warning about spacing out supplements and daily pills. The risk is invisible unless you know what to watch for. In some cases, calcium carbonate makes it hard for your body to get enough of certain antibiotics, especially tetracyclines and fluoroquinolones. Even iron and magnesium struggle to compete for absorption space if taken together with calcium carbonate.
Older people see the problem more often. Bones grow more fragile, more calcium comes in, and so do new prescriptions. Many seniors juggle a handful of pills before breakfast. This group faces a higher chance for unwanted effects, like heart rhythm issues or kidney stones, when mixing medication with calcium carbonate carelessly.
Chronic conditions pile up as the years pass. More doctors, more medications, and more potential for things to go wrong. Each change in routine may bring side effects that look like aging or the condition itself—when it’s really an overlooked interaction. I once helped an older neighbor sort her weekly pill organizer, learning she used a calcium antacid after breakfast but before her daily thyroid medication. After a check-in with her doctor, a two-hour gap between the two became a simple fix.
Most issues can get sorted out through simple timing tweaks. Space calcium carbonate two hours apart from other medicines that might react. Share your supplement routine openly at medical appointments. Pharmacists often spot trouble before doctors do, since they see the full picture across different prescriptions. Regular list reviews make the difference, especially after any new medication gets added to the mix.
People sometimes believe more supplements bring more health, but small details—like timing—play a bigger role than many of us imagine. Conversations matter. Your story, your routine, your body—these count more than generic advice or what worked for someone else. Careful combinations bring better results and fewer surprises.