People started paying real attention to Dimethyl Carbonate (DMC) around the late 20th century, when the chemical industry grew hungry for greener, less toxic solvents and reagents. Phosgene ruled the carbonate-making process for decades, but those who worked with it knew all too well about its dangers. The switch toward DMC brought cleaner air to labs and factories, with fewer headaches over regulatory compliance and emergency response. DMC’s production hit its stride after researchers nailed down less hazardous synthesis routes, mostly baking it from methanol and carbon dioxide with clever catalysts. That shift reflected a wave of regulatory pressure for safer, more sustainable alternatives, which in turn steered investments and academic work toward easy-to-handle substances for both technical and consumer markets. To this day, DMC keeps finding its way into new research settings, as it isn’t shackled by the same baggage as many older chemicals with dark environmental legacies.
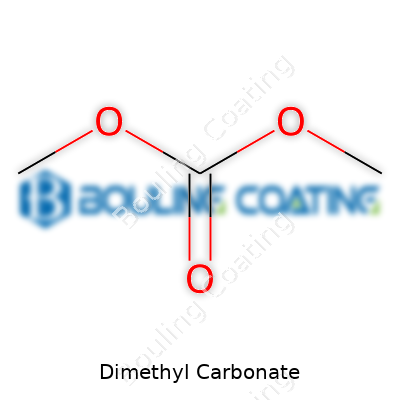
Dimethyl Carbonate stands out as a colorless, flammable liquid carrying a faintly fruity smell. Folks know it as an organic carbonate, but that label doesn’t do justice to its versatility. Some call it by other names—methyl carbonate or carbonic acid dimethyl ester pop up in older journals and chemical catalogs. Under the hood, DMC’s structure lets it play roles that other carbonates or solvents just can’t handle, making it the choice for anyone needing a low-toxicity, environmentally lean compound in the lab or on the production floor.
Nothing in the properties of Dimethyl Carbonate makes it especially challenging in day-to-day handling. With a boiling point around 90°C, it evaporates neatly without forcing complex engineering in manufacturing plants. DMC’s relatively high flash point helps keep workplace risk in check compared to more volatile solvents. It dissolves both in water and a wide range of organic liquids, which allows real flexibility—particularly if you’re formulating coatings, resins, or battery electrolytes. Its molecular weight of about 90 g/mol and density a little below water may seem trivial details, but in the real world of industrial scale-up, these numbers make batching, transfer, and storage tasks more straightforward, trimming time off setups or cleanouts. You don’t get those small wins from heavier, fussier liquids.
Older methods of making DMC poured on the phosgene and accepted the waste and toxic byproducts, which nobody would recommend today. Over years of research and tighter regulations, the top prep methods pivoted to transesterification of ethylene carbonate with methanol or the direct catalytic synthesis from methanol and carbon dioxide. Both push the reaction yields up and waste streams down. That carbon dioxide method especially grabbed attention in sustainability circles since it recycles a common greenhouse gas into a useful product. Researchers keep hunting for better catalysts—zinc, copper, or complex organometallics—hoping for higher selectivities, lower energy costs, and fewer headaches during post-reaction purification. In the academic world, these improvements mean a real chance to teach students about merging green chemistry with practical output, not just classroom ideals.
The fun with Dimethyl Carbonate doesn’t stop at simply using it as a solvent or intermediate. Chemists rely on DMC’s dual behavior: acting either as a methylating or a carbonylating agent. Reactions involving DMC yield everything from polycarbonates right up to advanced pharmaceuticals. For instance, methylating phenols or amines used to require tough, toxic reagents; DMC pulls off the same trick with less risk to both worker health and plant safety. It’s the go-to for making diphenyl carbonate—a key step in greener polymer production—and for making battery-grade electrolytes. In the lab, the wide reaction range draws both academic and industrial teams, since DMC’s milder conditions and workup simplicity slash both costs and hazards, proving that technical progress can serve the bottom line and safety targets at once.
Safety takes center stage anytime flammable liquids enter the picture. DMC’s relatively benign toxicity means it doesn’t burden users with the worst of the warning labels, yet its flammability keeps safety protocols tight. This isn’t the kind of compound anyone would keep near open flames or ungrounded electrical wiring. Respirable vapors and liquid splashes remain a concern; handling requires proper personal protective gear, robust ventilation, and storage in suitable vessels. Both European and North American agencies issue guidance that mirrors best industrial practices, not just bureaucratic checkboxing. In workspaces where DMC gets transferred outside closed pipes, mandatory training on spills or fires ensures that rookie mistakes don’t escalate to major damages or injuries. One overlooked point in regulatory talk: DMC’s low acute toxicity lures some into underestimating chronic exposure effects, so periodic air quality checks pay off in long-term workforce health.
The world’s EV makers, paint formulating chemists, and pharmaceutical process engineers can all open their supply room doors and find a drum of Dimethyl Carbonate in the lineup. Its low toxicity and light environmental footprint compared to older solvents win it a place in reformulation projects that chase regulatory deadlines. Lithium-ion batteries use DMC in electrolyte blends thanks to high electrochemical stability and sharp conductivity—in other words, a boost to battery lifespan and performance. Paints and coatings often pivot to DMC to lower volatile organic compound emissions, especially when facing down bans or supply chain pivots caused by stricter environmental rules. In drug manufacturing, DMC smooths the path for methylation and carbonylation steps, side-stepping the need for phosgene or dimethyl sulfate—compounds infamous for toxicity and regulatory hurdles.
Research on Dimethyl Carbonate never stopped at ‘good enough.’ Chemists spend enormous lab hours uncovering new catalytic processes, with the dream of running production lines at ambient pressures and temperatures. Process engineers take findings from academic projects and adapt them for scale, hunting for cheaper, less specialized catalysts and minimizing complex separation or recycling steps. Materials scientists keep stretching DMC’s reach into batteries, polymers, and specialty chemicals. Environmental scientists jump into the act by quantifying lifecycle emissions, since regulatory credits and corporate social responsibility hinge on real data. The drive to use carbon dioxide as a feedstock rather than as an emission links DMC research to climate strategy in a way few other basic chemicals can boast. Each incremental improvement helps not just the specialty chemicals field, but sets examples for greener, scalable plant operations.
Concern about chemical toxicity stays front-of-mind for any organization handling hundreds of liters at a go. Compared to a swath of traditional solvents, DMC keeps surprising folks with its relatively low acute and chronic toxicity profiles. Inhalation or skin exposure calls for vigilance, but evidence points to fewer severe outcomes than old standbys like toluene or acetone. Researchers track metabolites and long-term effects to guard against nasty surprises, and up to now, results mostly line up with established safety thresholds. It’s not a chemical without risk, but for a solvent that meets production volumes seen in batteries, binders, and coatings, it reads as a win in balancing performance and worker safety—not just regulatory box-ticking.
Dimethyl Carbonate sits in a crossroads of industrial change and sustainability ambition. Companies face growing pressure to lower their products’ environmental footprints while matching technical performance and output scale. DMC’s value as a drop-in for dirtier solvents and as a direct link to greenhouse gas reclamation raises its profile as both an economic tool and a climate asset. More governments and industry leaders recognize the need for greener synthesis pathways, so future work aims for not just incremental gains but a reinvention of chemical production chains. Success depends on collaboration across disciplines—chemists working with process engineers, environmental experts teaming up with plant managers—all chasing safer, higher-volume outputs with less waste and lower total lifecycle impacts. If this pattern continues, younger generations entering the field will likely know DMC less as a revolutionary product and more as a standard bearer of what modern, responsible chemical manufacturing can achieve.
Dimethyl carbonate rarely gets a headline, but it shapes industries and daily life more than most people realize. This clear liquid comes with a mild smell and plays a big role in how modern manufacturing keeps moving. Some describe it as a “green” solvent, but that doesn’t mean much until you see where it ends up: paint thinners, electronics, cars, and the packaging that surrounds the things we buy.
Walk into any factory where coatings and paint are made, and you’ll spot drums labeled with time-worn names, many of them old-school solvents known for their impact on air and water. Dimethyl carbonate gives these factories a new tool—one that cuts down on the dangerous volatile organic compounds (VOCs) that traditional petroleum-based solvents release. By swapping out harsher chemicals like toluene or acetone, workers get a safer space, and nearby rivers and parks benefit too. Studies show the U.S. EPA recognizes dimethyl carbonate as a safer substitute in certain applications, offering lower toxicity and easier handling compared to long-standing alternatives.
Anyone with an electric vehicle has a real stake in the story of this material. Dimethyl carbonate stands as a main ingredient in the electrolyte mixtures used in lithium-ion batteries—those same batteries that send phones, laptops, and cars zipping along each day. Why? It holds up well under charge and discharge, and it forms a stable “film” on lithium anodes, making those batteries last longer and catch fire less often. On the shelf, battery packs may look high-tech, but each owes a quiet debt to compounds like this one.
Shoppers who care about what goes into plastics also have reason to pay attention. Dimethyl carbonate brings flexibility to making polycarbonate plastics, which show up everywhere from eyeglass lenses to water bottles and CDs. Older ways of making these plastics often used phosgene—a chemical with a deadly track record. Dimethyl carbonate offers a cleaner route that leaves behind fewer toxic byproducts, and skips the need for corrosive or hazardous substances.
Car design keeps changing, but worries about air pollution stick around. Dimethyl carbonate gives fuel engineers a way to boost the oxygen content of gasoline, helping it burn cleaner and release fewer smog-causing compounds. Major research centers in Europe and Asia have tested blends of gasoline with small amounts of this compound, with the goal of tightening emissions standards and improving air quality in crowded cities. Switching over isn’t as simple as dumping in a new additive, but progress often starts with these small shifts.
None of these uses fix modern life’s problems overnight, but each stands as proof that change happens bit by bit. Producers, regulators, and engineers can steer more industries toward safer chemicals—if good options exist and governments commit to clear rules. Smart investments in research speed up the pace, and practical trials in real-world products help people see the benefits for themselves. As more companies adopt safer alternatives and drop the old, legacy chemicals, both public health and the planet win out.
Dimethyl carbonate isn’t flashy, but its impact runs deep, making the world a little cleaner with each new innovation it touches.
Plenty of chemicals get labeled "green" or "eco-friendly" the moment they're less toxic than something else. Dimethyl carbonate (DMC) has won praise in both academia and business for filling that niche as a solvent, a fuel additive, and a carbonate source. I keep hearing folks call it a breakthrough for sustainability—some even say it's the perfect substitute for old-school, nasty compounds like phosgene or methyl chloride. But words like "clean" and "safe" deserve some unpacking.
DMC breaks down quicker than a lot of the chemicals it replaces. That's real progress. In water and air, it doesn’t linger or pile up the way legacy chemicals do. If you spill methylene chloride, you’re looking at contaminated groundwater and a whole lot of regulatory headaches. DMC brings none of the same baggage, at least on the environmental persistence front.
Still, every process comes with tradeoffs. Most DMC production methods avoid highly toxic reagents, which definitely cuts risks to workers and communities nearby. Some modern factories rely on carbon monoxide and methanol, both bad actors themselves, but still a step toward using lower-impact chemistry. The really ambitious methods make DMC by reacting carbon dioxide with methanol. This creates an outlet for CO2 and could, if scaled right, help slow the rise of greenhouse gases. That’s one place DMC punches above its weight—removing a bit of CO2 while kicking older, more dangerous chemicals to the curb.
If factories run on coal power, even the greenest process spits out a big carbon footprint. DMC plants hooked up to renewables or low-emission grids can claim stronger environmental credentials. Sadly, much of today’s industrial chemistry still leans on fossil fuels, especially outside wealthy countries. DMC wins points in the lab, but production-side emissions still count for a lot.
Practicality matters, too. I’ve seen DMC replacing tougher chemicals in paints and plastics, and that’s good for air in the shop, but it doesn’t magically make a finished product biodegradable or non-toxic. Burning DMC as part of gasoline can cut some engine emissions, but it's no silver bullet. Environmental friendliness comes in degrees—not absolutes.
I’ve talked to manufacturing teams who say they’ll switch to DMC for worker safety alone—they’d rather store barrels of DMC than phosgene, any day. Medical research suggests DMC brings lower acute toxicity. Given how many jobs involve handling solvents, these small shifts add up. From an occupational health perspective, DMC moves the dial.
No chemical solves every part of a system in isolation. DMC's upgrades work best when paired with renewable energy, greenhouse gas capture, and mindful regulation. Demanding better tracking of energy sources and supply chains would help. Then there’s the problem of what happens after DMC leaves the factory—government pressure for green design and end-of-life recovery would keep momentum going.
Dimethyl carbonate represents a clear improvement over the old tools, no doubt. But calling it environmentally friendly without conditions feels optimistic. It’s a better answer, not the final one.
Most people outside of labs or manufacturing plants seldom cross paths with dimethyl carbonate. This clear liquid comes up in work with coatings, solvents, and the production of polymers or pharmaceuticals. I've seen how even experienced workers can treat chemicals as just another box on the checklist, but habits like that open the door to trouble. News about chemical injuries rarely names dimethyl carbonate, yet that doesn’t make it harmless. People get hurt from simple mistakes, not just from the scary substances we hear about all the time. This chemical smells like methanol and feels slick, but quiet danger lurks in its fumes and contact points.
I remember a young technician who splashed a little on his bare forearm, thinking, “It just feels cold, maybe a little dry.” He shrugged it off, but his skin let him know a few hours later that not every liquid is as gentle as water. Redness, slight burning—mild for him, but a sharp reminder that chemical exposure often sneaks up slowly instead of roaring in right away. Dimethyl carbonate irritates skin and eyes. In a job where you move fast and reach for beakers or bottles a hundred times a day, simple gloves and goggles start to feel like a joke. Yet one incident is enough to prove their value.
No one likes coughing fits or headaches at work. Folks familiar with organic solvents know the drowsy, spacey feeling that comes from an overexposure. This chemical evaporates easily, turning small spills or open containers into invisible hazards. Solvent vapors gather close to the ground, and if the room doesn’t circulate enough air, workers start suffering. Stories from colleagues back up the official advice—crack a window, run the exhaust, and keep even small jobs in the hood or ventilated area. People sometimes underestimate fumes from clear liquids, but your body knows the truth before your mind catches up.
Take a look at the label on a drum; that flammable symbol matters. Even if the liquid seems calm, its vapors catch fire much easier than water. Sparks from static or an electrical switch can do more harm than an open flame. One spill on a bench, a spark, and suddenly everyone in the room is scrambling. Choosing flame-resistant coats, grounding equipment, and cleaning up spills fast cut down on the odds of a scare. Only those who have cleaned up after a fire know how quickly neglect becomes regret.
People working around chemicals often do routine checks with half their attention somewhere else. Missing cracked hoses or loose lids doesn’t seem urgent—until a leak turns into a sprint for eye wash or the exit. I've found, through plenty of long shifts, that it’s cheaper and safer to take five minutes to fix a container than to call a site cleanup team. Nobody brags about the spill that didn’t happen—yet those are the real victories in any workplace.
Every major incident starts small. Habits spread across staff: checking protective gear, labeling bottles, asking questions when something feels off. Management can bring in training, run drills, and invest in better ventilation. Side-stepping shortcuts helps more than any manual does. People can push back against carelessness by listening to their senses and remembering close calls from the past. In the end, common respect and a little diligence shield us better than any regulation could.
Out in the world of chemicals, numbers on a chart often get tossed around like footballs. Still, boiling and melting points, especially for dimethyl carbonate, tell a bigger story. If you work in a lab or a factory, those numbers can decide how smoothly your day goes. Heat a chemical too much, and you risk nasty vapors or even a runaway reaction. Let it freeze, and you lose hours waiting for a block of solid to thaw. It's the sort of detail that makes or breaks a shift, makes chemistry either your friend or your nightmare.
Dimethyl carbonate catches the eye because it doesn’t bring the baggage of some harsh solvents: its reputation for being less toxic than usual suspects like methylene chloride gives it an edge in industrial and lab settings. That alone gets the attention of folks trying to keep things green and safe. But that reputation only gets you so far. You still have to work with it in the real world, which means boiling and melting points aren’t just trivia—they're a guidebook.
The scientific literature points to a melting point near 2°C (about 36°F). It doesn’t take much cold for dimethyl carbonate to go solid. If you keep your lab at fridge temperature, you’re probably pulling the bottle out and shaking it in hopes it'll liquefy. On the other end, the boiling point lands at 90°C (roughly 194°F). That’s hot but nowhere near the boiling point of water. That kind of range makes dimethyl carbonate accessible for folks with basic equipment, maybe with just a hot-plate and some ice in a cooler.
Drawing from my own days clocking hours in the lab, dimethyl carbonate always showed up as the flexible helper in reaction setups. The low melting point made storage a pain in cold winters, so the walk to the fridge turned into a guessing game. If the bottle got slushy, we’d run it gently under warm water, hoping for the best. The boiling point, not too high, meant gentle vapor came off fast, which called for solid fume hoods or tight lids. The stuff never set off smoke alarms, but nobody wants to breathe in more chemicals than necessary.
Chemists and engineers picking their solvents need numbers they can trust. Those melting and boiling points set the boundaries. With dimethyl carbonate, the narrow margin between room temperature and freezing can trip up an unprepared shift. I watched colleagues scramble with glassware gone frosty, the liquid gone milky overnight. Most chemical storerooms in big plants now use temperature monitors, catching those cold snaps before bottles turn slushy. It's a simple fix but takes commitment from the top down.
Lab safety doesn’t just mean handling spills or donning goggles; it means knowing your gear and watching your thermometer. Vapors from dimethyl carbonate, kicking off around 90°C, demand proper ventilation. Forgetting that gets risky, not just for your health, but for your project results. As more teams choose greener alternatives to harsh solvents, the practical limits of dimethyl carbonate need honest headlines, not marketing spin.
Choosing a chemical isn’t just about environmental badges. It's about lining up all the facts—boiling points, melting points, toxicity, and storage quirks—and weighing what fits the job. Newcomers and old hands alike benefit from learning why a bottle sometimes pours and sometimes needs coaxing just to flow. That kind of street-smart chemistry moves labs and workplaces toward solutions that work in the real world. Dimethyl carbonate offers plenty, but the science under those temperatures keeps honest folks cautious—and helps keep chemistry moving forward without unnecessary setbacks.
Handling chemicals like dimethyl carbonate often looks routine in a well-lit lab or a busy plant. But anyone who's seen a spill or a near-miss knows how quickly things can go sideways without the right setup. Years in manufacturing taught me that shortcuts with chemicals catch up in the end. With dimethyl carbonate, the smooth, clear liquid seems harmless enough—until it’s not. It carries flammable vapors, triggers irritation, and can eat away at some materials if ignored.
I’ve watched operators dump dimethyl carbonate into just about every type of barrel you can imagine. Only stainless steel or carbon steel drums, with tight-fitting, clean seals, stand up. The plastic barrels, no matter how tough they look, don’t always fare well with this solvent. Fitting the wrong lid or using a degraded gasket turns a drum into a leaky hazard. It also means you can end up breathing in the sweet, irritating fumes without realizing it at first.
Heat and this chemical don’t get along. The flash point sits lower than room temperature in some seasons; even a small bump from a sunny window or forgotten storage shed could push vapors over the edge. Keeping storage cool, shaded, and away from sparks or open flames is worth the extra step. I learned to double-check storage away from boilers, forklifts, and anything hot. Fans that pull air out, not just swirl it around, cut down risks fast.
All it takes is one rough road to turn careless packing into a crisis. Vans and trucks loaded up with dimethyl carbonate need proper anchoring. Metal containers should ride upright, with spill trays below. Any slosh or drum-roll can loosen caps, especially with temperature swings jarring the seals. Drivers need training too—no guessing about what’s in the back or fumbling with paperwork during traffic stops. Placards and documentation work as both warning and legal requirement, and police or responders move fast when they spot real labels instead of mystery tanks.
Once I saw a crew try to wipe out a dimethyl carbonate puddle with kitchen towels. A proper spill kit, with pads that actually soak up solvents and not just water, makes the difference. Fire extinguishers rated for chemical fires go hand-in-hand with these kits, just like gloves and goggles belong right next to every drum. Ventilation and storage away from oxidizers or acids finishes the safety chain. Forget those steps, and risk another round of the same old avoidable stories.
Some folks in management talk up regulations like paperwork hurdles. Out on the floor, those rules protect lives. Dimethyl carbonate counts as a hazardous material during shipping all over the world. Tracking every jug and drum, using only certified containers, and keeping a tight inventory matters for both legal and safety reasons. Re-training sessions and open reporting build confidence across teams—and catch mistakes before they end up on the news.
Handling dimethyl carbonate comes down to respect—for the chemical, for the people around you, and for the communities nearby. Treating storage and transport as more than just a box to check off cuts down on surprises nobody wants. The right gear, training, and attitude mean more than any safety slogan. That approach keeps workers healthy, regulators satisfied, and operations running long after the day's work ends.