Ethyl acetate has roots that stretch back to the 18th century, a time when curiosity about the chemical world inspired practical experiments in small European labs. Originally produced by distilling ethanol with acetic acid, chemists uncovered its characteristic fruity odor and seemed to marvel at the success in isolating an entirely new substance. For generations, ethyl acetate retained importance as an accessible solvent, long before modern industrial chemistry ramped up demand. During the 20th century, as synthetic chemistry advanced and manufacturing scaled up, new and more efficient methods of production appeared, eventually powering entire industries with ready access to this compound.
In daily life, ethyl acetate often goes unnoticed, yet it's in fingerprints left behind from nail polish removers, it's behind the sweet smell in certain paints, and it lingers in the air from many household products. The compound bridges gaps between chemistry labs and retail shelves. Chemists favor it for being volatile and effective at dissolving a range of substances. From pharmaceuticals to confections, its fingerprints can be found practically everywhere, often hiding behind generic-sounding ingredient lists. Looking at any chemistry storeroom, just about every shelf has a container of ethyl acetate within easy reach.
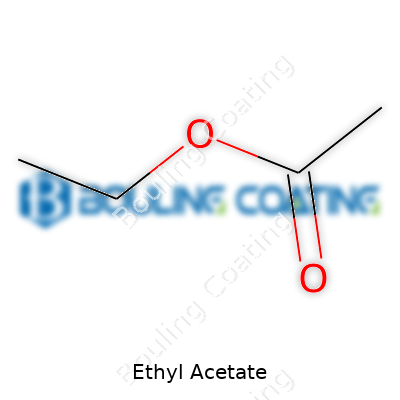
Pour a little ethyl acetate into a dish and you’ll catch a strong, sweet scent—reminiscent of pears or overripe fruit—which says a lot about its volatility. This colorless liquid evaporates quickly, flashes at relatively low temperatures, and dissolves easily in most organic solvents, but resists mixing with water. The low boiling point spells both convenience and caution; it speeds up drying in paints and coatings but also lends itself to easy inhalation, making ventilation especially important. The molecular formula, C4H8O2, reveals a simple ester organization. Chemists trust its reactivity enough to use it in high-energy reactions; the beauty comes from its ability to participate in or withstand a surprising number of chemical environments.
Boxes and bottles bearing ethyl acetate labels present a certain sense of familiarity in any laboratory or paint shop. From my own experience, those labels usually highlight high purity levels, often above 99 percent, given how contaminants could interfere with particular uses. Regulatory requirements insist on hazard pictograms, flammability signals, and instructions for safe storage. While some technical details can seem intimidating, the bottom line is straightforward: ethyl acetate is flammable and needs handling with care, while long-term storage away from direct light and heat ensures the chemical stays stable. For anyone mixing paints or preparing chromatographic runs, these details aren’t just paperwork—they’re assurance the solvent will perform as expected.
Traditionally, small batches of ethyl acetate come from combining ethanol and acetic acid in the presence of a strong acid, such as sulfuric acid. Industrial plants scale up this reaction, using recycled catalysts and refined separation techniques to crank out thousands of liters every day. Direct esterification still makes sense thanks to its relative simplicity and the steady demand for both starting materials. Larger facilities sometimes use alternative approaches, such as the Tishchenko reaction, especially where price volatility creeps in for feedstocks. After the reactions wind down, distillation removes water and unreacted materials, leaving behind a product pure enough for everything from glues to green chemistry research.
Chemists in universities and industry have used ethyl acetate to coax all kinds of transformations from starting materials. Its most well-known use in reactions comes as a solvent, and it makes life easier for hundreds of organic transformations, including Grignard reactions and extractions. Certain modifications to the ester bond itself hold interest for those in polymer or materials science; hydrolysis, for example, can break ethyl acetate down to acetic acid and ethanol, two chemicals that find use in their own right. Nitration or chlorination of ethyl acetate makes way for specialty chemicals, especially where high purity is critical. People who work with this compound value its willingness to stay out of the way—unless they want it to join the action and transform into something new.
As often happens in chemistry, ethyl acetate carries several other names around the globe and across industries. Some catalogs simply call it “acetic ether” or “ethyl ethanoate.” In the world of food science, its name appears in flavoring regulations and standards under terms like “banana oil,” reflecting that unmistakable aroma. For anyone needing clarity, the Chemical Abstracts Service (CAS) registry number offers a universal reference. These alternative names matter most in international trade, academic publishing, and regulatory filings, where misunderstanding can carry huge cost or safety consequences.
From the moment a drum of ethyl acetate lands in a factory loading dock or a lab supply room, everyone involved stays aware of safety rules. Proper ventilation, reliable flame-proof storage cabinets, and training all matter every bit as much as the chemical itself. Medical researchers and safety experts warn against chronic inhalation or extended skin exposure, referencing case studies linking such carelessness to headaches, irritation, or worse health complications. Fume hoods and gloves aren’t just fuss—they keep people safe day in and day out. Fire marshals around the world have published strict limits on how much of this liquid can be stored or transported at a time. For many businesses, these rules protect not just employees, but investments and neighbors. Growing up around paint shops, I learned quickly to respect the familiar red warning diamonds and always leave the area when the air started to take on a sharp, sweet scent during a busy shift.
Ethyl acetate covers a lot of territory in everyday products and manufacturing. Walk into any salon or hardware store and it quietly works behind the scenes in nail polish remover, lacquers, and adhesives. The printing industry relies on it for cleaning and ink formulation. Pharmaceuticals depend on its solvent abilities to purify and process ingredients. Winemakers can even detect ethyl acetate as a flavor note in spoiled wine. Laboratories around the world use it as a standard solvent for thin-layer or column chromatography—tasks that underlie drug discovery, material science, and environmental analysis. Even food scientists borrow it as a flavor enhancer, since in low amounts it boosts the aroma of many fruit flavors.
Research into ethyl acetate continues, driven by the push for greener processes and the hunt for safer, more sustainable solvents. Scientists take interest in bio-based approaches, attempting to ferment agricultural waste or surplus ethanol into high-purity ethyl acetate. Publications in green chemistry journals review catalysts that work at lower temperatures or produce fewer byproducts. Although mature, this industry still responds to evolving regulations on emissions and workplace exposure, so new methods frequently emerge. My own time in research labs showed me how the established protocols using ethyl acetate often formed the backbone of undergraduate teaching, reinforcing the value of safe, reproducible chemistry that doesn’t require rare or unpredictable materials.
The health effects of ethyl acetate continue to draw careful scrutiny. Decades of animal studies, workplace monitoring, and medical case reports provide a clear picture: while exposure to small, transient amounts produces little harm, concentrated vapors or repeated skin contact spell trouble. Regulatory agencies around the world, including OSHA and the EU, have set exposure ceilings based on measured thresholds of neurological, respiratory, and skin effects. A series of acute and chronic studies paint ethyl acetate as less dangerous than some solvents, but not something to take lightly. For those living or working near manufacturing sites, air quality standards offer some peace of mind. Still, the safest approach remains simple—good ventilation, protective gloves, and avoiding any unnecessary exposure, especially for children or those with breathing conditions.
Ethyl acetate carries momentum into the future as industries trend toward safer and more sustainable raw materials. Bio-based synthesis continues to gain traction, making use of plant and crop residuals in ways that lessen the environmental impact of massive solvent production. Demands from the electronics, coatings, and pharmaceutical sectors show no sign of tapering off soon. The rise in ecologically responsible business practices pushes producers to redesign processes to keep emissions low and byproducts non-hazardous. As regulatory requirements tighten worldwide, investment in cleaner, more efficient technologies looks like a smart bet. From what I’ve seen, demand for reliable, multi-purpose solvents never truly fades, and chemists appreciate materials that combine versatility with reliability. As innovation grows, ethyl acetate remains woven deeply into the fabric of everyday life and modern manufacturing.
Ethyl acetate turns up in a surprising number of places. I remember opening a can of paint thinner in my dad’s garage. That sharp, sweet smell always hit me first, even before I checked the label. This chemical finds life in everything from nail polish removers to the fruit flavor holding up your kid’s favorite candy. Factories lean on it for making products easier to dissolve, mix, or clean. Coffee without the buzz? Ethyl acetate cleans out caffeine in decaf without leaving strange flavors behind.
Industries trust ethyl acetate to get tough jobs done. Paints spread smoother and dry well because this chemical vaporizes quickly. Glue makers rely on it to dissolve sticky ingredients, securing the caps on every bottle. Printing companies see brighter colors in magazines and labels since it helps inks stick well to paper and plastic. Food labs can copy the taste of ripe pears or bananas, thanks to the fruity kick ethyl acetate brings.
More than personal products and food, this chemical carries weight in labs. During chromatography—one of those scientific tricks for separating mixtures—ethyl acetate helps researchers sift out the exact parts they need. It cuts through messes and leaves a clean split, leading to better results for researchers tracking everything from toxins to vitamins.
Ethyl acetate gets personal. It sits in perfumes, aftershaves, and anything giving off a “fresh” or “sweet” aroma. Growing up, my mom’s nail polish remover always filled the bathroom with a distinct hint of fruit and chemicals. That came from this very solvent, giving quick-dry properties while avoiding harsher chemicals found in earlier products. The chemical also plays a part in manufacturing flexible plastics and cleaning up electronics. A small drop can clean off sticky adhesives or old tape without much struggle.
People seldom notice it, but it keeps your ice cream and processed snacks tasting right. Ethyl acetate helps draw out natural flavors or build new ones without leaving behind aftertastes or harming health in small, controlled amounts. With the right hand guiding production and careful regulations, the food on grocery shelves can stay safe and enjoyable.
Any chemical that walks straight into homes and bodies deserves some scrutiny. Ethyl acetate evaporates into the air quickly, which cuts down on residue but does add to indoor air pollution. Too much exposure can irritate eyes, noses, or cause headaches. In the workplace, staff need solid air systems and good gloves. Public health agencies in the United States and Europe set limits to keep daily life safe. Now, industries look at cleaner production to cut pollution and recycle solvents, limiting waste from factories.
Switching to greener chemistry, tighter controls, and better recycling technology could shrink its impact. Safer alternatives sometimes cost more, but the long-term payback includes cleaner air, healthier employees, and fewer worries for families.
Ethyl acetate powers parts of daily living, from making treats taste right to keeping cars and houses looking sharp. Products get easier to use, companies stay productive, but health and environmental impact weigh heavily on how we handle chemicals at any scale. Trustworthy producers and watchful regulators keep everyone safer, giving us confidence in the air we breathe and the goods we put to work.
Walk into any lab and you’ll probably catch that sweet, sharp smell—ethyl acetate at work in cleaning, extraction, or chromatography. Students tote it through chemistry classes, painters wipe brushes with it, bakeries coax flavors because it dissolves naturally in fruits. Over the years, hands-on experience has forced me to respect this colorless liquid. I’ve seen newcomers splash it around, thinking it’s as harmless as table vinegar. A quick sniff soon teaches a different lesson. Ethyl acetate evaporates fast, and its fumes fill the air before you know it.
Breathing in those vapors too long isn’t smart. My own headache after working near open jars left no doubts. Studies back that up. At low exposure, most adults won’t get anything worse than mild irritation in the nose and throat. Keep splashing or soaking, chances rise for dizziness, drowsiness, or even nausea. It can irritate eyes on contact and dries out skin after repeated handling. Fire risks also deserve respect—the stuff ignites easily at room temperature.
Tracing guidelines from trusted agencies, the US Occupational Safety and Health Administration has set an exposure limit. Workers can encounter up to 400 parts per million over an eight-hour shift before those effects show up. That's a decent safety cushion, but crowded spraying rooms or labs with poor airflow quickly exceed this number if people aren’t paying attention.
People tend to skip gloves for quick cleaning jobs. The thinking goes, “It evaporates so quickly, how much could possibly get absorbed?” This underestimates the ability of organic solvents to work through the outer layer of skin, especially over a day full of repeated contact. Accidental splashes to the eyes hurt more than most expect, and washes with water might not be enough. Accidents happen faster than lectures can explain the risks. I've seen seasoned coworkers get a spill and end up with red, burning skin for hours.
The safety toolbox has plenty of answers. Simple fans pull vapors away. Disposable nitrile gloves make more sense than bare hands. Safety glasses or goggles turn bad splashes into minor annoyances. Storing small amounts away from heat keeps surprises at bay. Rags soaked with ethyl acetate can ignite without a spark. Something as simple as keeping them in sealed metal cans prevents disasters.
Nobody benefits from paranoia. With thoughtful ventilation and the right personal protective gear, risks drop low enough that businesses and labs keep ethyl acetate in daily rotation. It pays to read up, ask questions, and check one’s work area for airflow and cleanup supplies. Science says that handled with respect, ethyl acetate won’t cause harm. Complacency, not the chemical, brings trouble.
Ask a chemist to rattle off the boiling point of ethyl acetate, and most won’t miss a beat. 77.1 degrees Celsius. It sounds simple—just a number on a chart—but that number carries a lot of weight beyond the surface. Years working in research labs have a way of teaching you that knowing this isn’t only about passing exams or lab safety quizzes. It influences how we store, transport, and use tiny bottles and 200-liter drums of the stuff every day.
Ethyl acetate fills an odd niche. You’ll find it in nail polish remover, glues, and the extraction processes inside coffee decaffeination plants. What draws so many people to use it? This solvent doesn’t just dissolve compounds; it evaporates quickly thanks to its modest boiling point. That means chemists and manufacturers can recover other materials from mixtures without resorting to harsh heat—vital for keeping flavors, colors, or pharmaceuticals stable.
Watch hope flicker on the face of a young researcher prepping a chromatography run for the first time. Ethyl acetate’s lower boiling point compared to heavier solvents gives them flexibility and speed. Less energy consumption and gentler processing allow both high-throughput labs and small startups to keep costs in check.
It’s easy to treat boiling points as academic figures, but in practice, a few degrees higher or lower can turn a day bad. Too volatile and your solvent evaporates out of beakers, escape hood filters fill up, and the air in the lab gets thick with fumes. Many who have handled ethyl acetate recognize its fruity aroma—a warning you’re inhaling vapors that, over time, aren’t the best for your health.
Facility planners and plant managers can’t ignore such chemical traits. The boiling point hammers home the message that closed systems, ventilation, and fire prevention plans are not optional. Facilities that move a lot of ethyl acetate put explosion-proof refrigerators in play. Fire marshals get creative about placement of flammables cabinets. All these precautions draw straight back to how easily ethyl acetate slips from liquid to vapor.
Workplaces that handle ethyl acetate keep safety sheets pinned to the wall. Experienced operators understand that open containers and poorly labeled drums court disaster. Wear proper PPE and use even the smallest quantities in spaces with solid ventilation. Community firefighters train for scenarios with flash points well below water’s boiling mark.
The march for greener chemistry aims to weigh solvent choices against safety and environmental costs. Some researchers hunt for alternatives with higher boiling points or lower toxicity, but striking a balance isn’t always straightforward. Mainstream chemistry hasn’t abandoned ethyl acetate because of its versatility. Still, greener processes that cut down on solvent use or recover more of it before release see steady growth.
Knowing that ethyl acetate boils at 77.1 degrees Celsius isn’t just a tidbit for trivia night—it’s a touchstone for safe lab work, responsible manufacturing, and thoughtful innovation. Ignoring the practical sides of these data points always lands people in trouble, but taking them seriously opens up smarter, safer approaches for everyone involved.
Ethyl acetate slips into the background at many factories and labs, but this colorless liquid rarely causes trouble until storage gets sloppy. One whiff of the vapor leaves a sour tingle in your nose, and that’s not just an annoyance—high concentrations can knock ventilation workers off balance and, worse, turn a workplace into a fire trap. I’ve opened cabinets where the sharp smell hits hard, and that’s a sure sign someone let the cap loose or stored it where heat builds up. Fumes love to escape, and once they mix with air, even a small spark creates a real ignition risk.
People often forget how fast ethyl acetate moves from liquid to vapor. In warm rooms or near hot equipment, the rate climbs. This compound catches fire at temperatures lower than a summer midday, and that should put every storage decision under the microscope. I’ve seen stored solvents leak or evaporate through plastic, ruining labels and even shelving because the wrong type of container got used. Metal cans with tight-fitting lids solve much of that, and grounded safety cabinets give a double layer of defense against accidental ignition.
I’ve watched new lab workers catch headaches and dizziness — not realizing it came from poorly contained ethyl acetate, not just a long day. The amounts might seem tiny, but repeated exposure creates bigger problems over time. Hands get chapped from splashy handling. Storage inside eye-level cabinets puts people at risk every time they reach overhead. Eye injuries, skin burns, and persistent cough become workplace norms if storage drifts toward convenience rather than care.
Choice of room also plays a big role. Stacking a drum of ethyl acetate near oxidizers or acids can spiral into disaster, though people cut corners for space all the time. Local codes make sense here, not just as legal hoops but as lifesaving habits. Ground-level ventilated storerooms, away from direct sunlight, cut down on fume pressure and discourage quick evaporation. Staff who know to look for vapor leaks, or who can spot corroded shelves, catch small risks before they turn serious.
Storing chemicals comes with real-world consequences. Most accidents around ethyl acetate start with preventable mistakes—improper cans, clogged vents, or cabinets set too close to heat. Insurance claims rise fast after a fire or spill, but so does damage to reputation. In my years around shop floors, I’ve seen operations shut down for weeks and teams scrambling to explain how a single misplaced drum spiraled into a major loss.
Facility managers shouldn’t settle for the bare minimum. Installing flammable liquid cabinets helps, but regular checks and honest communication matter even more. Labels fade, and inventory logs often fall behind; regular walk-throughs and scheduled container inspections prevent the quiet spread of risk. Spills need quick, well-trained responses—no one should fumble for a plan or search for missing absorbent pads as vapors fill the room.
Ethyl acetate isn’t going away—it’s part of paint shops, R&D labs, and even flavor houses. Treating it like any nondescript solvent opens the door to serious harm. Training makes the biggest difference. Everyone—chemists, technicians, custodians—needs to spot trouble and fix it before accidents build up. Good storage practices are not a badge of bureaucracy but a real step toward safe, efficient work. Letting routines slip or taking shortcuts with a chemical so flammable never pays off in the long run.
Spend a day in a laboratory, a paint workshop, or any nail salon, and you’ll likely run into the sharp, fruity smell of ethyl acetate. It pops up in everything from glues to perfumes. People rely on it for its effectiveness—not just in dissolving things, but in making other substances mix more easily. That usefulness comes with a catch: ethyl acetate burns easily. Strike a match, let a vapored container go unchecked, or ignore proper storage, and the hazards become real, not just something you read about on a label.
I remember a colleague rushing into the break room at our shared workspace, wild-eyed after someone left a bottle uncapped near a heat source. The room filled with warnings, not flames, but that ten minutes of frantic airing-out stuck with everyone. The basic reality: this solvent can turn a minor oversight into an emergency. Even in outdoor spaces, vapors hover close to the ground and can drift toward an open flame or electrical spark, igniting faster than most folks expect.
Numbers don't lie. Ethyl acetate catches fire at temperatures far below boiling water—think summer afternoons, not industrial ovens. What stands out from safety bulletins and industry reports: a rapid flash fire can spread across a surface coated in a thin layer of this liquid long before anyone smells smoke. People handling ethyl acetate often underestimate how easy it is to reach those troublesome vapor concentrations in the air. In one incident at a local manufacturing shop, storage drums ended up too close to a faulty extension cord. The resulting fire, small yet costly, got traced right back to overlooked vapors from a leaky cap.
Big industries hammer home basic rules during training. Don't store ethyl acetate near heaters or direct sunlight. Use only in well-ventilated areas. Keep containers sealed tight. Few outside the chemical trade follow these habits at home or in casual use. It makes a difference. Even a rag soaked in the solvent becomes a hidden fire risk, especially if left bunched up in a trash bin. These aren't just safety slogans; they're direct responses to real accidents.
Tougher labels or flashy warnings will only help so much. People remember stories and shake-ups more than legalese. What works: regular reminders in workplaces, visible safety posters, and encouraging open chatter about close calls so nobody shrugs off risky shortcuts. Shops that use ethyl acetate regularly have locked flammable storage units, marked disposal bins, and staff who actually believe mishaps could hit their own bench. From what I've seen, this practical culture creates fewer emergencies than rooms stuffed with safety pamphlets.
Ethyl acetate proves useful until people ignore how fast it burns. Every bottle and jar should serve as a reminder: flammable doesn't mean maybe. Common sense beats regulations that sit buried in a drawer. Work with this solvent, and you know the drill—respect the risk, keep it contained, and never trust a sunny shelf or a half-closed cap to keep your space safe.