Methyl acrylate, with its sharp, pungent scent and colorless appearance, seems simple at first glance, but don’t let that fool you. This chemical, often overshadowed by its bigger cousins in the acrylic family, carries a history that winds through decades of industrial advances and scientific curiosity. Chemists began to unlock the secrets of acrylates in the early twentieth century, chasing new materials as plastics reshaped everyday life. Once laboratory-scale production grew into industrial batches, uses for methyl acrylate multiplied far beyond the original hopes. Factories around the world soon tapped into its ability to form plastics and resins. Looking back, widespread adoption did not spring from a eureka moment but grew steadily from trial, error, and real demand in coatings, adhesives, and textiles.
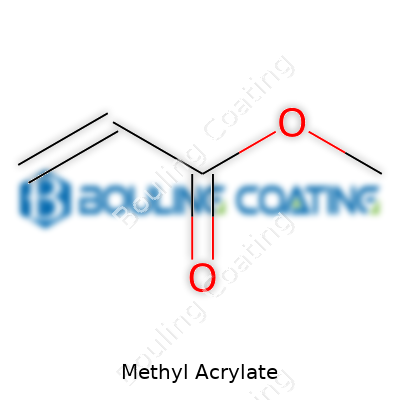
Chemists see methyl acrylate as a small ester molecule, formally known as the methyl ester of acrylic acid. The way it behaves—volatile yet reactive—gives it just the right mix of flexibility and utility for countless applications. Its formula, C4H6O2, speaks to its simplicity, but its physical properties set it apart from other acrylics. With a boiling point just above 80°C and a density below water’s, this liquid evaporates quickly if left open to air. The double bond in its structure invites polymerization, so it can form long chains when prompted by heat or light. This trait made methyl acrylate indispensable to industries seeking paints, sealants, or even superabsorbents.
In practical use, factory operators and safety managers know methyl acrylate for the exacting standards required to keep it both useful and under control. Its volatility means storage facilities need good ventilation and containers that won’t corrode or leak. Safety labeling warns of its flammability, its potential to irritate skin, and the need for protective gloves and eyewear. Labeling laws in many countries require standard warnings—flammable liquid, toxic if inhaled—based on real risks documented over years. No shortcut replaces good safety habits. Industries that work with methyl acrylate pay close attention to regulatory updates and technical bulletins, since requirements for labeling, packaging, and transportation have only grown tougher.
Years back, small-scale chemists relied on esterification, blending acrylic acid with methanol in the presence of sulfuric acid. These days, major manufacturers moved to processes that juggle purity, cost, and environmental impact. Careful control of temperature and reaction times stops runaway polymerization. Raw acrylic acid may come from the oxidation of propylene, tying methyl acrylate production to the fate of the petrochemical industry. The shift toward greener manufacturing—less sulfuric acid, less waste—nudges companies to review decades-old habits. Innovation doesn’t need a revolution; it grows from marginal gains in catalyst design, recycling of unreacted materials, and sharper monitoring of effluent streams.
Anyone who has mixed methyl acrylate with a strong base or a radical initiator sees a quick, sometimes violent response. Polymer chains form swiftly, and workers must stay vigilant. Methyl acrylate participates in copolymerization, linking with vinyl acetate, styrene, or acrylonitrile. This flexibility unlocks varied textures and strengths in the final materials—soft, tacky adhesives or tough, shatter-resistant plastics. Chemical engineers learned to modify methyl acrylate with functional groups that improve compatibility or tailor properties. In research labs, the molecule serves as a building block to test new crosslinkers, tackifiers, or surfactants, each step feeding back into the knowledge base used by industry.
Step into a warehouse or scroll through a chemical catalog and you’ll find synonyms like methyl 2-propenoate or acrylic acid methyl ester. The CAS number helps sort confusion, giving everyone a common reference. Industries and regulators know that an error in naming costs time and, at worst, safety. In my own experience reading technical datasheets, chemical identity ranks right up with handling instructions. Precision in naming practices—across languages and regulatory regions—cuts down on near misses and lost orders.
Long-term health of workers drives operational standards just as much as regulatory fines. Methyl acrylate presents dangers that earned it tough safety protocols. Studies on acute and chronic effects flagged irritation to the skin and eyes, respiratory troubles, and a headache-inducing vapor. Operators learn to spot leaks by smell but depend on gas detectors for certainty. Good plant design uses closed transfer systems, local ventilation, and regular air monitoring. Best practices evolved through real incidents—splash accidents, fires, and a few tragic losses. Safety depends less on paperwork and more on training, maintenance, and a culture where every worker can raise a flag if a process feels off.
Methyl acrylate’s versatility underlies its broad use. Take coatings—flexible films for cars, electronics, or furniture depend on the toughness and clarity it imparts. In adhesives, it gives that signature “snap” when a glue sets between two surfaces. Textile makers value softness and stretch, so methyl acrylate turns up in synthetic fibers blended into everyday clothing. In newer areas, researchers push boundaries—hydrogels for wound dressings or controlled-release systems in pharmaceuticals. What surprises me is how an unseen ingredient can shape both the durability of a commercial floor and the comfort of a bandage.
Universities and corporate labs never stop looking for the next edge. Green chemistry stands out—ways to cut down on emissions, lower energy use, and break dependence on fossil raw materials. Ode to open science: data on biodegradability, toxicity to aquatic life, and safer alternatives pile up in public databases, guiding companies before regulators have to step in. There’s a lively race to engineer better catalysts, both to boost yields and cut toxic byproducts. Some groups publish ways to integrate renewable feedstocks, turning agricultural waste or even CO2 into acrylic acid and, in turn, methyl acrylate. These advances sound far off but ripple into daily operations as soon as they prove reliable at scale.
Toxicology built up piece by piece, through careful studies in animals and real-world exposure incidents. Low-level exposure triggers coughs or skin redness in factory workers, but high exposures bring more severe symptoms: difficulty breathing, lasting eye injuries, or changes to nerves over time. Some research links methyl acrylate to DNA changes in cell cultures, though translating those findings to everyday health risks needs caution. Regulators in Europe, the US, and Asia sweep through the latest studies, balancing chemical utility against risk. The pattern in safety is clear: test, share information, improve process controls. My work on industry podcasts often circles back to this theme—open, honest reporting keeps people safer, even when the economic stakes run high.
Demand for methyl acrylate will not vanish. Substitutes exist in certain applications but fall short on cost or performance. What drives real change is pressure to lower emissions and waste, both from regulation and people working the production line. I’ve seen small tweaks in batch protocols add up to less exposure, fewer complaints, or cleaner wastewater. The next decade looks set for greener synthesis—bio-based routes, better recycling, and materials with lower hazard ratings that can match methyl acrylate’s reliability. Research into safe-by-design compounds may even nudge some applications in new directions, staking out a future where flexibility and responsibility can travel hand in hand. The communities that manufacture, use, and regulate methyl acrylate will need tough conversations about trade-offs, but the history of this molecule shows that progress—however gradual—follows a path set by necessity, creativity, and hard-won lessons from the shop floor.
Methyl acrylate might sound like something far removed from everyday living, but it comes up in all sorts of things you’d never expect. I only caught onto it by reading ingredient lists on products at home. Toothbrush handles, nail polishes, even certain glues in shoes often link back to this one chemical. Its real power is versatility. Chemists use it to make plastics tough, flexible, and clear. It isn’t glamorous, but the impact runs deep in regular life.
Factories rely on methyl acrylate to make something called polymers, which shape up into paints, adhesives, and special coatings. Looking at my own walls, I realize that water-resistant paint owes part of its strength to this compound. Same thing for the laminate on an old kitchen table. Manufacturers count on methyl acrylate to help coatings stick or to add some flexibility where brittleness might cause cracks. If you’ve ever pulled plastic wrap tightly over a bowl and liked how it clings, methyl acrylate helps in that design, too.
Nail salons use products that benefit from methyl acrylate because it cures hard and fast under UV light. The bottles don’t list it by name, but this chemical gives that satisfying, clear surface that won’t chip off from a gentle bump. Some folks in building trades may recognize its uses in floor polish, industrial coatings, or sealants that need durability, weather resistance, or gloss. It isn’t about luxury—builders simply want floorings or tools that last through cold, heat, and heavy use. Methyl acrylate steps up for those needs every day.
There’s a shadow to methyl acrylate. The stuff smells harsh. If you’ve ever been near a workshop making plastics or adhesives, you might catch a stinging odor. Breathing too much can cause throat or lung irritation. Long-term, high levels get even more serious, with studies pointing to risky effects over time. Having studied chemical safety myself, I know how vital simple habits like wearing gloves and working in well-ventilated areas are when handling any acrylates.
Regulations make a difference. Companies handling this material face restrictions. They monitor exposure levels, train workers about safe handling, and keep up with stricter industry standards. These regulations didn't appear out of thin air—they stem from real incidents and worker protections that demanded better oversight. Research keeps going, and groups like OSHA and NIOSH in the U.S. offer guidance to further reduce health risks. I find reassurance in how established chemical makers invest in training and safer process improvements, partly because of watchdog oversight and public demand.
The world of chemicals changes fast. Sustainable chemistry gets more attention now than it did a decade ago. Research focuses on finding alternatives that work as well, cost less, and pollute less. As consumers push for more eco-friendly products, companies start thinking about greener manufacturing, even for staples like methyl acrylate. I see these changes in the rise of water-based paints, more transparent labeling, and investment in recycling technologies.
In real life, understanding substances like methyl acrylate lets regular people ask smarter questions about what’s inside common products. We share a responsibility to demand safe workspaces, press for environmental responsibility, and keep an eye on innovations that promise something better in the long run.
Those who have ever spent time in a lab or worked around chemicals know this: some substances demand extra care, not only out of respect for the rules but because a wrong move can change everything. Methyl acrylate falls into that category. Used in resins, adhesives, and a range of industrial products, it brings the benefit of versatility, but it’s got a sharp edge: its vapors irritate eyes, skin, and the respiratory system, and it’s highly flammable.
The odor of methyl acrylate alone isn’t enough to give fair warning. Too many folks make the mistake of thinking they’ll smell harm coming, but the human nose hits its limit fast. After a few minutes, you can stop noticing the dangerous vapors and end up overexposed. Stories from old colleagues who got headaches or rashes after brief exposures still stick with me. Safety goggles and gloves aren’t about following codes; they protect your skin and eyes from immediate irritation and long-term harm.
Closed spaces and methyl acrylate simply don’t mix. Thinking you can “just open a window” leads to trouble. Fume hoods or proper exhaust systems keep the air breathable and reduce fire hazards. A fan in the corner won’t cut it. I’ve watched what happens when the ventilation slips, and the results aren’t pretty—burning eyes, coughing fits, work ground to a halt. Good airflow keeps the job moving and keeps people healthy.
It’s easy to get sloppy with routine. You start thinking you know every angle, until a spark reminds you how flammable methyl acrylate can be. Flammability isn’t just a technical detail—it’s a real risk to lives and livelihoods. Grounding containers, using non-sparking tools, and keeping the chemical away from ignition sources like hot equipment or open flames can’t be left to chance. Just last year, a fire broke out in a local plant when someone forgot about static from their plastic containers. The aftermath shut down production for days and put several lives at risk.
Lab coats, goggles, and nitrile gloves are basic, but some people still skip steps, cutting corners to save a few minutes. Every time, the same issues crop up: chemical burns, eye irritation, persistent rash. It’s not just about comfort—long-term or repeated exposure links to more serious health problems. In workplaces I’ve joined, the best safety records come from teams that treat these habits as second nature, not as an extra chore.
A well-organized chemical area is a line of defense. Storing methyl acrylate in airtight, labeled containers away from heat and sunlight keeps it stable and cuts down on risks. Leaky caps or forgotten open bottles have led to evacuations and sick coworkers more than once. Putting it on a high shelf with flammable warning signs can prevent small mistakes from turning into full-blown emergencies.
Shortcuts tend to show up most with new team members. Real-life demonstrations and hands-on training sessions, not just reading manuals, stick better. I’ve seen folks pay more attention after seeing a simulated incident or listening to a longtime worker’s real stories. The safety culture in a workplace grows out of these shared experiences, not from paperwork alone.
Protection from methyl acrylate isn’t about paranoia—it’s about knowing your environment and looking out for each other. Teams that keep their eyes open, gear up, and communicate well set a standard everyone benefits from. Safety works best when it’s woven into the daily routine, not forced at the last second.
Most folks outside the chemical industry don’t give a second thought to the ingredients that go into paint, adhesives, or even the plastics in everyday objects. Methyl acrylate tends to slide by unnoticed, though it quietly touches our lives more often than people expect. This compound shows up in everything from pressure-sensitive tapes to plastics that line food packaging. Methyl acrylate earns its place because of a small but mighty molecular structure that gives products their lasting power and flexibility.
Methyl acrylate’s chemical formula is C4H6O2. This isn’t just a bunch of letters and numbers. Each element—carbon, hydrogen, and oxygen—works together to make something useful. The structure starts with a simple framework: it’s an ester, built from acrylic acid and methanol. Think of it like a chain of carbon atoms, where one end clings to a double-bonded oxygen and the other wears a methoxy group (-COOCH3).
Chemists would sketch it as CH2=CHCOOCH3. That double bond between two carbons matters: it makes methyl acrylate reactive, letting it snap together with other simple molecules to form long chains—polymers. The ester group sitting at the end plays its own role, helping it blend into other resins or plasticizers. This elegant, simple structure almost seems designed for modern manufacturing.
In my own experience, the structure-specific story turns practical in a laboratory or workshop. That reactive double bond lets methyl acrylate build everything from soft contact lenses to water-resistant coatings. I remember working with a lab team, watching the polymerization process kick off as soon as we mixed the acrylate with a catalyst. The chemical structure led directly to the outcome: flexibility, stretch, sometimes sticky or smooth—there’s purpose behind every reaction.
The widespread use brings up a bigger conversation. This isn’t just about industry or chemistry textbooks. Exposure to methyl acrylate can cause headaches, eye irritation, and other health effects. The simple structure that fuels its usefulness also means it can evaporate quickly. I’ve seen plant operators suit up in protective gear, fully aware of the risks tied to its volatility. Handling and ventilation are big concerns. The U.S. Environmental Protection Agency lists methyl acrylate as a hazardous air pollutant, so keeping emissions in check isn’t just red tape—it’s necessary for worker safety and community health.
There’s always room to push for safer alternatives or improved designs. Some manufacturers have started using enclosed reactors and advanced scrubbing systems to trap emissions and recycle raw material vapors. Labs now experiment with new catalysts to make the polymerization process cleaner and more efficient, pulling down energy use and waste. Big improvements show up when health and safety officers work directly with research staff. As someone with a foot in both worlds, I see the value in honest collaboration over just ticking off compliance boxes.
For anyone curious about how chemistry intersects with daily life, methyl acrylate draws a pretty clear path. Knowing its formula and structure goes beyond trivia—it lays out both the possibilities and the responsibilities that come with handling these industrial building blocks. Being mindful doesn’t get in the way of progress; it keeps people engaged, alive, and ready for tomorrow’s challenges.
Methyl acrylate doesn’t show up in most people’s conversations, but the way this chemical gets handled matters a lot. It’s the sort of stuff that can help create everyday items like adhesives or plastics, yet its sour, pungent odor should be a warning. The risks are not theoretical. It can irritate eyes, skin, and lungs—sometimes badly. There’s no room for shortcuts with storage and transportation. From working in an industrial setting, I’ve seen what happens when care gets replaced by hurry. That’s why clear protocols and honest attention to the details make all the difference.
A barrel of methyl acrylate looks harmless sitting in a warehouse, but its flammability and toxic vapors are hiding in plain sight. Temperature control makes or breaks this effort; it can polymerize if it gets too warm, leading to dangerous pressure build-up. Workers don’t want to deal with burst drums or wrenched chemical lines, so keeping it cool—ideally below 30°C—really matters. That’s not some abstract best practice; it’s survival.
Real-life experience tells you another thing: always use dry and well-ventilated storage. The fumes creep up on people, especially when containers aren’t sealed tightly. Simple steps, like keeping containers away from direct sunlight and heat sources, save headaches and trouble. Fire sprays, spills, and emergency showers belong nearby. Chemistry doesn’t cut corners, and neither should workers or facility managers.
I’ve met truck drivers proud of getting a tough shipment through rain, snow, or heat. Hauling methyl acrylate means taking extra steps. Vehicles must be checked for leaks, seals, and material compatibility. Containers built to handle strong chemicals aren’t a luxury—they are the right way to avoid accidents on highways. Imagine a minor collision turning into a chemical spill. That’s frightening, and it happens more often than news outlets report.
Regulations like those from the Department of Transportation in the US and ADR in Europe exist for good reason. They lay out how chemicals travel, including the labeling, placarding, and documentation. This isn’t just bureaucratic red tape; a clearly marked tanker or drum reduces confusion for fire crews and emergency teams if trouble hits. Anyone who has witnessed an emergency response knows fast, correct info saves lives.
Digital monitoring tools for temperature and humidity inside storage buildings give real-time feedback. Alerts prevent small issues from turning into real disasters. Training workers, not just giving them a manual to sign, pays off too. I remember a safety officer running hands-on drills—afterward, people stopped skipping steps. That’s where trust in a company’s safety culture gets built, not just written down for inspectors.
It all comes down to accountability. If workplace leadership values safety over speed, the ripple effect gets people watching for signs of leaks, keeping exits clear, and double-checking paperwork. Shortcuts belong nowhere near hazardous chemicals. This isn’t about a checklist; it’s about facing the real risks head-on, with both brains and integrity running the show. The public never sees these behind-the-scenes decisions, but the impact goes far beyond the walls of any one company.
Most people never hear the words “methyl acrylate” unless they work in chemical plants, paint production, or certain plastics manufacturing. This clear liquid might seem harmless, but plenty of research warns otherwise. The real issue comes from how it can sneak into lungs, skin, or eyes during work. I spent some time in industrial settings, and stories about sudden headaches or rashes get traded almost as much as sports news. Those conversations aren’t for nothing.
Methyl acrylate puts the respiratory system at risk. People exposed on the job sometimes talk about sore throats, coughing fits, or feeling short of breath. The vapor carries a sharp, irritating smell, which is no surprise since even brief exposure can sting the nose or chest. Experience in the paint shop taught me that chemical headaches or eye watering often mean poor ventilation, not a random cold. Fail to manage air flow and people start calling in sick more often.
This chemical isn’t any kinder to skin or eyes. A splash can trigger redness, itching, or worse—blistering burns. Even small droplets can start a rash, especially after hours on the floor without proper gloves. Eye exposure tends to hit fast and hard, causing pain and temporary vision trouble. Some of my old coworkers learned this lesson after hurried cleanup work. Lab studies have tracked allergic skin responses, proving these reactions aren’t just rare flukes.
Short exposures bring discomfort, but the long haul presents more serious concerns. Chronic exposure to methyl acrylate links back to asthma-like symptoms and ongoing respiratory issues. There’s growing discussion in occupational medicine circles about chemical sensitization, where even modest amounts can set off the immune system after repeated contact. Some animal studies suggest methyl acrylate may carry cancer risks, but evidence in humans looks less certain.
The body’s metabolism breaks down methyl acrylate into substances that can stress the organs. In large doses, these byproducts overwhelm the liver and kidneys. The nervous system takes a hit too, and reports of dizziness or tiredness aren’t just coincidence.
No single fix will erase these dangers, but smart steps cut risks down. Proper training, airtight gloves, and eye protection make a difference. Shops need crisp ventilation and up-to-date air monitors. Workplaces should also run regular health checks and rotate duties when possible, to limit repeated exposure. Sharing personal stories around these safety steps helps too—after all, people listen to coworkers more than they read wall posters.
Policy plays its part as well. Rules set by health agencies call for strict exposure limits, and for good reason. Stronger enforcement and quick reporting help keep companies honest. On the front line, workers who speak up fast save themselves and their teams a world of trouble.
Real protection comes from small habits and solid culture, not from pretending that these risks sit in distant factories. It takes attention, honesty about symptoms, and a shared commitment to safer routines. This keeps methyl acrylate from turning up as the reason someone can’t work, or worse, can’t recover.