The chemical world tends to remember its useful compounds through the industries they change. Ethylene glycol ethyl ether acetate came out of early 20th-century research chasing effective, safe solvents. As the paint, ink, and coating businesses looked for alternatives that balanced evaporation rate, solvency, and lower flammability, this ether ester stood out. Companies gravitated toward it because the unique blend of the glycol ether backbone and acetate tail seemed to solve quite a few problems. Over the years, the story of this material has followed broader trends—less hazardous workplace environments, tighter air-quality rules, and the global shift away from some of the older, more toxic chemical solvents. Chemists and engineers didn’t just stumble onto this compound—they built its utility out of careful bench work and a deepening understanding of both synthesis and safety.
It’s easy to overlook a clear, colorless liquid, but ethylene glycol ethyl ether acetate stands out for doing exactly what it promises. This solvent shares much with its ether relatives: mild odor, high boiling point, relatively low volatility. But the ethyl ether acetate side helps it straddle the line between “water-miscible” and “organic.” In practical use, that means painters, ink makers, and electronics technicians appreciate how readily it dissolves stubborn resins, acrylics, and nitrocellulose. It isn’t as aggressive as some ketones and it tends to dry a bit slower, supporting smoother finishes and less risk of “blushing” in coatings. These properties, combined with miscibility in many common solvents, keep it flexible. Everyday details matter—a boiling point usually near 156°C, a flash point that allows for safe storage below room temperature, and decent stability on the shelf.
Clear, honest labeling carries extra weight with solvents like this. Safety Data Sheets don’t just check a regulatory box. Workers trust those numbers with their skin, eyes, and lungs day in and day out. In my time working around industrial coatings, the tiniest deviations in labeling caused confusion. With ethylene glycol ethyl ether acetate, regulations in markets like the US, EU, and Asia tie its product labels to tight purity ranges and require strict notation of hazards—including specifics about flammability, exposure limits, and emergency protocols. These standards reflect hard-won lessons from factory floors and shipping docks. And while product codes and supplier names fill out the paperwork, it’s the rigorous definitions from groups like OSHA, REACH, and local agencies that keep this solvent safer to buy, store, and use.
The underlying chemistry behind ethylene glycol ethyl ether acetate is a classic case of “building up” from smaller ingredients. Chemists start with ethylene oxide and ethanol, using them to make ethylene glycol ethyl ether, which is then reacted with acetic acid or acetic anhydride. The route matters—control the temperature too loosely or let moisture slip in, and purity drops fast. Down the line, small tweaks to the standard preparation method yield products better suited for certain coatings or inks. Once made, this compound plays nicely in many reactions, thanks to its inertness under most application conditions. Its ability to dissolve tough resins nods to the balanced relationship between polarity and molecular size. The option to tweak, recycle, or safely dispose of this solvent marks another reason it has stuck around for decades, outlasting many rivals that ran afoul of changing regulations or worker safety concerns.
Anyone who’s dealt with chemical supply knows the names multiply far beyond what you see in textbooks. Ethylene Glycol Ethyl Ether Acetate shows up under names like 2-Ethoxyethyl Acetate, EGEEA, or even “Cellosolve Acetate” in older documents. This mix of IUPAC and trade names means anyone handling procurement or inventory has to double-check what actually sits in the drum. Research papers, regulatory filings, and shipping manifests reflect these varied synonyms—making it all the more important to rely on CAS numbers and exact chemical identifiers. My own headaches with old stockrooms taught me never to trust only a trade name or casual abbreviation; mistakes can range from minor delays to major safety hazards.
Chemicals like this teach respect in the workplace. While not as acutely dangerous as some other solvents, ethylene glycol ethyl ether acetate carries enough risk to deserve gloves, goggles, and solid ventilation. Repeated exposure may lead to headaches or skin irritation, and the chemical has implications for reproductive health with chronic overuse. National occupational exposure limits reflect these threats. I’ve seen how training and clear labeling help lab techs and painters treat solvents with caution, not fear. That extra step—a fume hood, a well-fitted respirator—stops accidents before they ever become a line in a safety report. Every drum, every bottle, needs periodic inspection. Leaks, vapor buildup, and old residue add up fast if ignored. Focusing on these standards in daily practice supports longevity for workers and end-users alike.
Markets rely on ethylene glycol ethyl ether acetate in more ways than most people realize. From cleaning up printing rollers to thinning complex lacquer systems used in automotive plants, the compound shows up in spots where lesser solvents just can’t handle the load. I’ve watched electronics manufacturing rooms lean on it when prepping or cleaning sensitive parts without swelling or dissolving plastics. Art conservationists sometimes turn to it for cleaning age-old paintings or removing stubborn varnishes—gentle enough when handled right but powerful enough to lift years’ worth of residue. Even though new regulations push companies to explore lower-emission or “green” alternatives, the reliability and predictable performance of this compound keeps it on many tech and production lines worldwide.
Academic labs and big R&D centers spend resources on solvents because improvements cascade through entire industries. Over the last decade, investigations into ethylene glycol ethyl ether acetate have dug deep into two major questions: how to tweak its molecule to deliver safer, less toxic derivatives, and how to fine-tune performance for the next generation of polymers, coatings, and films. Teams run studies on vapor emissions, waste treatment, and environmental breakdown. Data from environmental monitoring links spills and long-term contamination with the wider “Forever Chemicals” debate, fueling work to make both use and disposal safer. Real advances happen where scientists, manufacturers, and policymakers meet—everyone from senior chemists down to the warehouse stevedores has a story about what could go wrong if you turn your back on basic safety or lose track of a product’s physical and chemical quirks.
Ask any health and safety specialist and you’ll hear real concern. Ethylene glycol ethyl ether acetate, while less notorious than some glycol ethers, doesn’t get a free pass. Decades of industrial monitoring tie chronic exposure to effects on blood counts and reproductive health. That’s not just distant theory—I’ve worked with teams sorting through legacy factory safety reports and realized the importance of regular monitoring, from proper handling to keeping break rooms solvent-free. Field studies show how improper storage or poor PPE policies open the door for accidental inhalation or skin absorption. Researchers spend time testing air samples, examining breakdown metabolites in urine, and working with animal models to get ahead of any long-term damage. Safer alternatives continue to get their day in the lab, but for now, firm boundaries and clear worksite training matter more than ever.
Years of debate over chemical safety and environmental sustainability shape what happens next for ethylene glycol ethyl ether acetate. Regulatory agencies set tighter standards on emission rates, update exposure limits, and encourage greener practices. Industry researchers keep looking at molecular modification to lower toxicity or boost recyclability. Every time I walk a factory floor or read new legislation, it becomes clear—companies can’t lean forever on legacy chemicals without adapting practices and products. The next breakthrough may come from green chemistry—maybe new catalysts or renewable feedstocks will nudge this and similar compounds toward lower impact. As more people worry about indoor air quality, workplace wellness, and environmental footprints, chemical manufacturers face rising expectations to prove both safety and performance. In the meantime, the industry continues to balance staying competitive with keeping an eye on worker health, regulatory obligations, and environmental responsibility.
Step inside a modern electronics factory, and the ordinary eye would miss the chemical forces keeping things moving smoothly. Behind the glossy shell of a smartphone or the subtle sheen of car paint, Ethylene Glycol Ethyl Ether Acetate—often called EGEEA—plays a quiet but essential part. This colorless liquid seems unremarkable, but manufacturers lean on it for good reason.
In my years covering industry developments, I’ve seen chemicals like EGEEA show up where high performance counts. Its main calling card is acting as a solvent. You might not see it splashed across labels at the hardware store, yet it brings real benefits in coatings, inks, and cleaners. Its structure, marked by a balance of volatility and solvency, strikes a careful note that helps paints flow on smoothly and printers deliver sharp, durable images.
Electronics can't live without precise cleaning and delicate application of coatings. As device makers chase ever-thinner components and brighter screens, residues become the enemy. EGEEA cuts through greasy or sticky residues, clearing the microscopic pathways that signals travel through. Its ability to dissolve difficult substances—like certain resins—gives chip and display makers flexibility when building new devices.
Paints and varnishes rely on this same power for an even, dust-free finish. Whether the goal is a scratch-resistant dashboard in your car or a spatter-free mural sprayed on a city wall, EGEEA keeps pigments and binders dispersed. Workers apply coatings at all temperatures—even in sticky summer heat—because it keeps drying times workable. Households or auto shops often notice longer-lasting, glossier surfaces when formulations use the right solvent mix.
Coverage of solvents often stops at their performance, but real world use always has a flip side. In communities dotted with manufacturing plants, residents worry about chemicals like EGEEA escaping into the air or water. Health researchers pay attention to its vapor, since inhaling too much can irritate the eyes, nose, and lungs. Workers on paint lines or inside printing plants need regular training and solid ventilation to avoid exposure.
Lawmakers and local agencies keep pushing for greener chemistry. As I’ve seen in discussions with industry leaders, many are looking for safer alternatives or better containment measures. Some companies redesign products to use less solvent or to switch to water-based systems. These moves cost time and money, but they pay off with lower emissions and fewer health complaints among workers.
Manufacturing rarely stands still. Every year brings new gadgets and cleaner production standards, all connected through tough conversations and choices about materials. Regulators, factories, and workers engage in a running dialogue about what risks are worth it and what protections matter most.
Ethylene Glycol Ethyl Ether Acetate doesn’t grab headlines. Yet its behind-the-scenes role shows how everyday items—electronics, paints, coatings—depend on chemicals that have both perks and pitfalls. Better monitoring and smarter engineering hold promise, along with the willingness among industry to rise to new challenges. Watching these changes happen up close, one thing feels certain: the future depends on understanding what these hidden helpers do, and how we interact with them.
Ethylene glycol ethyl ether acetate, also called EGEEA, shows up often in paint shops, printing presses, and electronics factories. It might not cause alarm at first glance — it’s a clear liquid that smells slightly sweet, almost like a weak perfume. I’ve handled it myself while cleaning ink rollers in a printing workshop, and there’s a quiet danger in how ordinary it looks and smells. This is exactly why many workers, especially those just starting out, ignore the risks.
EGEEA soaks into skin far too easily. After my first day cleaning trays with no gloves, I noticed redness on my knuckles and a strange tingling sensation that didn’t fade for hours. Days later, a more experienced co-worker pulled me aside and told me stories of headaches, nausea, and dizziness spreading through the room after someone spilled a batch without proper ventilation. The chemical’s reputation for damaging the nerves and kidneys isn’t just theory. The United States Environmental Protection Agency points out that it can affect the nervous system after just a few hours of breathing in heavy vapors. Reports also link long-term exposure to reproductive issues and slow kidney damage.
Most workplaces throw around the term “PPE” — personal protective equipment — almost like a buzzword. EGEEA isn’t a chemical you want to approach with half-measures. Industrial-grade nitrile gloves, chemical splash goggles, and heavy aprons stop direct contact. Standard surgical gloves won’t help, nor will regular safety glasses, since the vapors slip past them. Long sleeves and chemical-resistant shoes keep you out of trouble. A friend of mine ran a small customs painting shop and once thought a few open windows would handle the fumes; the next day, every painter was coughing and squinting. Mechanical exhaust fans saved the day, filtering and diluting the air faster than open doors ever could.
One habit that cuts injuries down fast involves basic hygiene. Workers can’t just wipe chemicals off — they need to wash exposed spots with lots of soap and water, not just sanitizer. I always tell new hires to treat break rooms as shoe-free zones, since traces carried in on boots or gloves eventually find their way onto sandwiches and coffee mugs.
Storing EGEEA in tightly sealed containers, kept away from heat, keeps the risk low. I once watched a new manager stack chemical drums in direct sunlight behind the shop; two days later, the tops began to bulge. Flammable vapors build up quickly here. Keeping storage rooms cool and dry with clear hazard markings around them gives everyone time to react if something leaks or combusts.
Chemicals like EGEEA don’t need to be mysterious. Straightforward training, without the jargon, sticks better. Anyone allowed to handle the liquid ought to see how to put on a mask, how to read the Safety Data Sheet, and where the nearest spill kit waits in case of trouble. Mock spill drills might sound boring, but the first real emergency always feels less scary if your hands already know what to do.
All these steps sound basic, but skipping even one brings real danger. EGEEA doesn’t forgive carelessness, and stories from the shop floor prove that more than any training video or manual ever will.
Plenty of folks probably walk by chemical names on packaging without a second glance. One glance at “Ethylene Glycol Ethyl Ether Acetate”—and a head starts spinning. These formulas get tucked into the fine print on paint cans, cleaning supplies, and industrial products. Even if most eyes glaze over, these details can carry more weight than most notice. Knowing the chemical formula and its CAS number isn’t only for lab techs and chemists. It's key for safety, compliance, and making sure what's inside a product matches what’s promised.
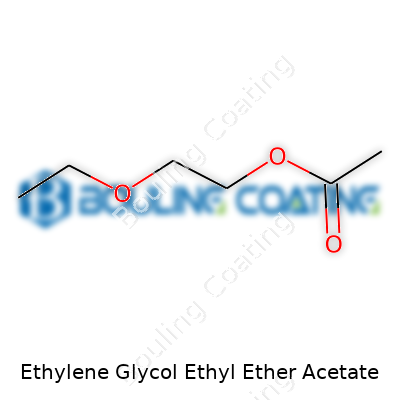
Ethylene Glycol Ethyl Ether Acetate has the chemical formula C6H12O3. Plugging this into a search brings up a world of papers and safety sheets, but it’s that string of numbers—111-15-9—known as the CAS number, that traces the identity of this compound in every database worldwide. These numbers keep everyone on the same page, from plant workers to government regulators, so there’s no mixup with similar-sounding chemicals.
During one summer working in an industrial coatings plant, those squiggly chemical names and numbers started to feel less like jargon and more like trusted markers. Chemical drums rolled in by the pallet, each marked with technical language. Only with the right formula and CAS number could staff match up what vendors claimed with what safely belonged on the mixing line. If you’ve ever seen a spill or an unexpected smell on the floor, you know the panic that sets in when details on a drum don’t match up with what’s supposed to be inside.
Beyond the warehouse, these identifiers drive global trade. Customs inspectors use the CAS number to clear shipments. Techs at recycling plants track which drums are safe to salvage and which demand protective gear. The rules for import, storage, and disposal hinge on these specifics. Companies face fines—or worse—when they mishandle chemicals because someone guessed or made assumptions.
Plain language chemical information arms folks with the knowledge to make real choices—about what they put in products, what they store on-site, or which supplies belong in the school science closet. Mislabeled goods can lead to fires, health risks, and even environmental disasters. Several chemical incidents, from minor workplace exposures up to large-scale leaks, start with simple confusion or a lack of clear labeling.
For people who work with chemicals, even those without technical backgrounds, knowing the formula for Ethylene Glycol Ethyl Ether Acetate—C6H12O3—and its CAS number 111-15-9 helps cut through confusion. Honest, visible details give workers, neighbors, and regulators a fighting chance to keep themselves and their communities safe.
Industry leaders have a responsibility to use accurate and consistent identifiers. Training programs can go beyond the safety basics by teaching teams how to read chemical labels and cross-check them in public databases. Everyday workers, not only chemists, keep an eye out for new shipments, strange odors, and odd labeling. Companies that foster open communication about chemical content see fewer mishaps.
It comes down to respect—for science, for regulations, and, most importantly, for the people who interact with chemical products daily. The chain of safety starts with naming things right. If every product label carried clear formulas and the right CAS number, workplaces and homes would have fewer accidents and more trust.
Anyone who’s spent time around chemical supply rooms picks up on a simple truth early: the little details around chemical storage can decide between a regular day and a disaster. Ethylene Glycol Ethyl Ether Acetate is one of those substances often tucked onto a middle shelf, thought of as routine. That’s a mistake. Overlooking its quirks can put people, property, and the local ecosystem at risk.
In my years shadowing chemical technicians and environmental engineers, I witnessed plenty of storage closets that didn’t inspire confidence. Too often, folks assume factory-issued containers and a closed door give enough protection. Yet Ethylene Glycol Ethyl Ether Acetate responds poorly to heat, light, and careless handling. Containers can warp, seals break down, and then vapors sneak out.
Places with fluctuating temperatures make the risks worse. A humid spring can bring subtle corrosion, followed by a dry, brittle fall that makes containers crack. One month the label looks new, the next it’s smudged and unreadable. In the end, nothing replaces hands-on vigilance—checking seals, reading labels, inspecting for leaks, and making sure nothing’s too close to a vent or heater.
People sometimes underestimate just how fast vapor concentrations rise in a closed space. Lab techs often rely on their sense of smell or guess based on past experience, but Ethylene Glycol Ethyl Ether Acetate doesn't always give clear warnings. Prolonged exposure carries real health risks. That puts a burden of care on anyone handling or storing it. Ventilation is non-negotiable in any room where solvents live. A fume hood or an outdoor shed works better than a closet in the corner of a warehouse.
Fire safety matters just as much. I’ve seen solvents stacked near electrical panels or extension cords draped through rooms packed with containers. Static electricity may seem harmless, yet one accidental spark can be enough. Grounded storage racks and avoiding open flames can save more than a few headaches.
A shelf is more than a place to put a bottle. Quality shelving won’t rust easily. Closed cabinets, especially those designed to hold flammables, offer better protection. Clear signage with chemical names and hazard triangles doesn’t just look organized—it buys critical seconds if somebody ever needs to grab the right bottle fast. Locked cabinets mean only those with training get access. I’ve found that small steps like this cut down on mystery spills and lost containers.
Too many times, unused or forgotten chemicals pile up in the corner. Over time, separated layers form inside old bottles, and caps get brittle. That’s not just wasteful. Any chemical stored too long becomes a question mark—does it behave the same, or has its risk profile changed? Regular inventory checks and a habit of dating containers help. Getting rid of waste according to actual regulations is smart stewardship, not just following the law. Less clutter means fewer hiding places for danger.
Storing Ethylene Glycol Ethyl Ether Acetate with respect shows care for people working alongside you and everyone downstream in the supply chain or the neighborhood. It’s about responsibility, not routine. Getting it right keeps workspaces safer and communities healthier. Sometimes, it’s the things we overlook that most demand our attention.
Every so often, a chemical with a technical name like Ethylene Glycol Ethyl Ether Acetate makes its way out of industrial circles and lands in public conversations. Known in laboratories and factories, this solvent gets used in paints, coatings, inks, and cleaning agents. Its job often involves breaking things down, thinning materials, or helping stuff to dry more evenly. I have spent time in industrial settings and have seen workers handle drums labeled with a long list of safety warnings — Ethylene Glycol Ethyl Ether Acetate features on those lists.
Anyone working with this chemical quickly learns about its risks. Exposure can happen through breathing in vapors, letting droplets touch your skin, or — rarely, but riskily — accidental ingestion. From what I’ve seen and read in safety training and chemical hazard databases, even a little contact can irritate the eyes, nose, and throat. Factory workers sometimes complain about headaches, dizziness, or fatigue after a few hours near open containers. The underlying problem is this: Ethylene Glycol Ethyl Ether Acetate doesn’t just evaporate harmlessly like water. It gets into the air and, with enough of it, can travel through the lungs and blood, affecting multiple organs.
Scientific studies have linked heavy or long-term exposure with more serious health problems such as damage to the liver or kidneys. Some research from regulatory agencies warns about potential reproductive risks if levels stay high for long periods. The fact is, few people outside of factories face chronic exposure, but those who do risk facing real health problems unless proper safety procedures stay in place.
Environmental impact forms another piece of the puzzle. Ethylene Glycol Ethyl Ether Acetate doesn’t just disappear after pouring it down the drain. In local rivers, it has the potential to harm fish and other aquatic life. It breaks down slowly, especially if large quantities get unleashed at once, and this gives it time to spread in water and soil. Air pollution remains a problem as well. Vapors contribute to the formation of ground-level ozone, and communities living near large factories sometimes complain about strong odors or headaches traced back to solvent use.
The big takeaway from these facts points to one clear step: control. Smart companies and careful workers use protective gloves, goggles, and masks. Good ventilation in factories makes a visible difference. I remember watching an air quality monitor spike whenever someone opened a can without extraction fans running. Spills cleaned up right away don’t leave as much mess in drains or storm sewers.
Governments keep a close watch too. Limits on workplace exposure, requirements for proper disposal, and permits for emissions help keep communities safer. Switching to less hazardous chemicals where possible makes a lot of sense. Safer alternatives exist for many applications. Even small changes, like substituting water-based paints for solvent-based ones in home use, reduce overall risk.
Those with experience in industry know that not every solvent can be easily replaced, but clear labeling, worker training, and tight controls go a long way. Keeping health and environmental risks from Ethylene Glycol Ethyl Ether Acetate in check takes cooperation between employers, regulators, and workers on the ground. Transparency about chemical hazards strengthens trust and helps everyone breathe a bit easier — sometimes literally.