Isobutanol isn’t new on the chemical scene. You can trace its origins as a commercial product back to breakthroughs in fermentation processes and petrochemical refining during the twentieth century. Chemists noticed that not all alcohols act the same way—a subtle change in molecular shape or synthesis route can lead to very different outcomes on the plant floor or in the real world. Isobutanol grew out of that realization. Its main production took off during the rise of industrial chemistry, when every extra step in synthesis had to justify itself with usefulness and cost savings. I’ve seen how those old research notes, listing limits of possible alcohols, gave rise to today’s specialty solvents and fuel additives.
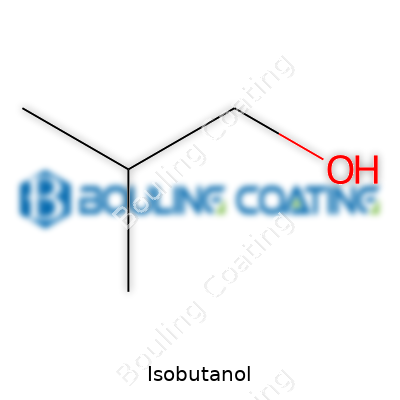
Isobutanol belongs to the family of four-carbon alcohols, with a branching that sets it apart from its straight-chain cousin, n-butanol. The molecule’s branching looks simple—one methyl group forks off from the main chain—but that shape gives it properties that fit fuel compositions, specialty coatings, and even pharmaceuticals. People often confuse names like 2-methyl-1-propanol, i-butyl alcohol, or even its EINECS number, but all of these refer to that same versatile liquid. I’ve come across bottles labeled in all sorts of ways, yet the distinctive aroma and the way it blends into solutions don’t change.
On paper, isobutanol checks in as a clear, colorless liquid with a mild, alcoholic smell—not much different in appearance from the vodka in a freezer, but don’t mistake this lab staple for something you’d want in a drink. It burns with a blue flame, boils around 108 degrees Celsius, and carries moderate polarity. In practice, that means it slips into both water and hydrocarbon mixes, cutting through grease or forming solutions that ethanol can’t quite handle. That balance plays a real role in paint strippers, glues, and even as a fuel blendstock—applications that exploit its higher energy density and lower hygroscopicity than ethanol. I’ve watched as fuel engineers tinker with ratios to get the stability they need for modern engines or shelf-ready chemicals.
Labels on drums and containers might describe assay levels, water content, storage requirements, and handling standards straight from GHS or OSHA regulations. In real-world terms, this means tight seals, stable temperatures, and a whole lot of respect for the fumes. Vapors build up quickly in poorly ventilated spaces, and concentrated spills demand gloves and goggles. Safety sheets urge caution, but real safety relies on routine: regular leak checks, vapors monitored, and everyone trained well on what to do if things go sideways. I’ve sat in those safety briefings where the old hands swap stories about minor slip-ups that became big learning moments.
Most isobutanol today still comes from petrochemical refineries, using oxo synthesis that couples propylene and synthesis gas with a carefully chosen catalyst. Fermentation from renewable feedstocks like corn or sugar beet sits at the edge of market expansion, driven by biotech innovations that rewire microbial metabolism to crank out more isobutanol than yeast would make naturally. I’ve followed the debate over which method wins—old fossil-based chemistry or newer, greener bioprocesses—the answer always comes down to cost swings, regulatory changes, and what kind of feedstock price swings hit that quarter. No one recipe dominates forever, and pilot plants quietly tweak conditions so that yields and purity climb a little with every run.
Isobutanol’s molecular shape gives it reactivity spots different from that of n-butanol. It takes up halogens or acids at predictable spots, yielding esters, ethers, or other derivatives found in flavors, fragrances, and fuel additives. The primary alcohol group stands up to most mild oxidizers, but vigorous reactions can break it down to isobutyric acid. I’ve watched R&D teams dig into these chemistry quirks, hoping for that one catalytic shortcut that saves hours in the pilot reactor or opens a door to a new product line not crowded with competition.
Pick up a catalog or speak with chemical suppliers, and you’ll find isobutanol under names like 2-methylpropan-1-ol, i-BuOH, or sometimes just isobutyl alcohol. Trade names pop up too, but in the end, it’s that structure—three carbons in a chain, a methyl branch, and an alcohol group—that sets the rules for how it behaves, no matter which label gets slapped on the drum. Names matter for shipping, customs, and old habits, but the applications come back to that same molecular flexibility.
Safe handling goes well beyond reading container labels or checking off items on a safety audit. In the lab or plant, routine ventilation and proper personal protective equipment stop acute exposure before it starts. Headaches and dizziness serve as early signals—no one ignores these if they’ve worked around organic solvents much. Storage means watching out for open flames, sparks, or static electricity—fuel-rich vapors mix readily with air, raising the risk of flash fires. Regulatory updates change recommended workplace limits for airborne concentrations or spill protocols, so companies and workers often update their practices to avoid incidents that can harm people or shut down production. These days, stricter environmental limits nudge facilities to implement tougher emission controls, even if the gear comes at a cost.
You might walk past isobutanol’s impact every day without noticing. Major uses pop up in paints and coatings, where it balances evaporation rates and helps solubilize resins for smooth finishes. Fuel blending engineers appreciate its low volatility and resistance to water pickup, a crucial advantage over ethanol in humid climates or legacy storage tanks. In the world of plastics, pharmaceuticals, or extractive chemistry, isobutanol steps in as a dependable solvent—sometimes overshadowed by flashier or newer molecules, but almost always somewhere in the mix. I’ve had my hands stained with lacquer thinners that owe their bite and long working time to a healthy dose of isobutanol.
Research groups look past routine uses, hunting for improvements in manufacturing routes, environmental sustainability, and downstream product innovation. Genetic engineering now tweaks microorganisms to turn waste agricultural streams into high-purity isobutanol, tempting manufacturers with visions of lower carbon footprints and fewer price shocks from oil markets. Analytical chemists hunt for minor impurities that can trip up sensitive processes or new applications that need even tighter purity specs. Process engineers push for continuous production lines that cut costs and energy use, gradually raising the bar on what “state-of-the-art” means. In university labs, students test isobutanol as a platform for next-generation fuel cells or bio-based solvents, eager to carve out new commercial fields.
Toxicologists dig deep into how repeated exposure hits the body, flagging the nervous system effects that show up at higher concentrations. Short bursts—like an accidental whiff—usually pass, but chronic exposure can erode health, especially in poorly ventilated settings. Animal studies suggest moderate risk, though at doses much higher than regular workplace exposures. Public health researchers look for links between workplace exposure and long-term illness, while regulators track cumulative emissions in air and water. The discussion now includes so-called “green chemistry” initiatives, which push for solvents that offer the necessary performance while reducing persistent environmental risks and replacing more toxic alternatives, like heavier alcohols or glycol ethers.
Talk to a few industry veterans, and you’ll hear predictions that isobutanol’s day is far from over. Demand as a fuel extender grows in regions that want lower volatility, fewer emissions, and engines that last longer between tune-ups. Paints and coatings still rely on that subtle balance between evaporation and solvency, which means alternatives would have to prove themselves on a crowded shelf. Bio-based isobutanol sits at the edge of commercial viability, championed by start-ups and farm co-ops alike, encouraged by carbon reduction targets and renewable fuel standards. Challenges remain: costs swing with market instability, and public skepticism follows any new process promising to “green” the chemical industry. Competitors like methanol or ethanol will always have a cost advantage, but they can’t always meet the technical demands that isobutanol handles easily. Having watched how the field evolves with every generation of chemists, I see no sign that isobutanol will be relegated to the sidelines. Its precise chemistry, evolving production, and flexible uses keep it in the game.
Plenty of folks might glance at the name “isobutanol” and think it’s just another tongue-twisting compound meant for labs and manufacturing plants. The reality looks a bit different. Isobutanol steps out of the factory quietly and makes its way into a pile of everyday things, changing how products work and how we live without much fanfare.
The most familiar role for isobutanol comes through the gas pump. Many drivers don’t realize some of their regular fuel might already have isobutanol blended in. Ethanol, long known for stretching out gasoline supplies, grabbed that headline—yet isobutanol brings its own punch. It packs a higher energy content per gallon than ethanol and stays stable when stored, even with water around. Adding isobutanol to gasoline helps reduce greenhouse gas emissions, which matters in the bigger fight against climate change. For folks like me who care about what our tailpipes put into the air, seeing isobutanol getting used as an advanced biofuel brings a bit of hope.
Factories lean on isobutanol for more than fuel. Think about the paint rolled onto your living room wall. Isobutanol helps paints flow better and dry just right. It preserves workability so the color slides on evenly, without turning gummy. The same goes for coatings on furniture, cars, and electronics. Beyond paint, isobutanol steps into creating plastics, rubber, and even cleaning products. Its versatility lets product makers tweak formulas instead of scrapping them, so they aren’t left scrambling for alternatives every time regulations or costs shift.
For years, most isobutanol came from fossil fuels. Companies now increasingly turn to microbes and fermentation tanks to make it from renewable plant matter. I’ve watched enough news about droughts and forest fires to know our fuel and chemical supply chains can’t lean on fossil sources forever. Bio-based isobutanol keeps the doors open for an industry trying to lighten its waste and carbon footprint. That said, building up enough supply of renewable isobutanol means new investment, smarter crops, and more efficient ways of turning sugars into finished product. Nobody has this perfectly figured out, though steady progress pushes things forward.
Switching to isobutanol doesn’t always mean trading away safety. The stuff still demands respect—breathing its fumes or letting it soak into your skin can cause harm. Regulations kick in to keep exposures in check around people and wildlife. For paints and cleaning products, newer green solvents and natural oils give manufacturers another angle, cutting down possible health risks tied to stronger chemicals. Each new breakthrough usually takes time to show if it really works on a large scale or just piles up other issues, so science and regulation need patience as well as urgency.
In my view, isobutanol won’t ever turn into a household word, but its benefits run deeper than most folks realize. From lowering emissions at the pump, to making manufacturing smarter, to blending bio-based solutions into the products we use, isobutanol plays a valuable part. Still, moving forward means paying attention: cleaner production, smarter regulation, and alternatives that balance progress with safety for people and the planet.
If you’ve ever worked inside a chemical lab or watched someone hand off a clear liquid from a bulk drum, you know people get lax with routine. Isobutanol looks a lot like many harmless solvents, but you’d regret treating it as such. Years ago, I witnessed a guy carry an open bottle across the bench, chatting away, and he barely missed sending half a liter splashing across his clothes. Lucky for him, he wore a lab coat. Not so lucky, he spent the afternoon with burning skin and a pounding headache. Turns out, those “small mistakes” leave a sting.
Isobutanol evaporates easily and the fumes have teeth. Inhaling them can feel like a punch to your lungs, bringing headaches or dizziness. Skin picks it up fast, too: typical irritation, redness, even blistering if you get complacent. Some folks might think it’s alright to take their gloves off just to fill a pipette “for a second.” Wrong move. Keep those nitrile or butyl gloves on until you’re completely done and double-check for clean hands before you grab your phone, lunch, or the stapler.
Anyone who’s caught chemical splatter to the face won’t forget it. Isobutanol in your eyes feels like rubbing sandpaper along your eyelid. Reaching for rinse water becomes your only mission. Even a drop can leave you blinking in pain for days. Those goofy goggles by the door sit there for a reason. Wear them even if you think you won’t be at the bench long. Isobutanol won’t care about your schedule.
Tucking isobutanol away in a random cabinet can lead to trouble. Like other alcohols, its flash point means even a small spark can torch the air above a spilled drop. No one plans for lab fires—they just happen when bottles tip or get left near heat sources. Grounded metal safety cans and flame-proof storage do more work than you think to keep surprises at bay. Routine checks for leaks and labeling beats sorting through a melt-down aftermath.
You don’t want to rely on the “sniff test” for isobutanol—its scent creeps up slowly. Good fume hoods and active ventilation keep your breathing air cleaner. Remember, if your nose picks up that sharp, sweetish odor, the levels may already be pushing safe limits. Open windows do less than you think. Make sure the ventilation is built for chemistry, not just comfort.
Practice matters. People freeze when accidents happen unless they’ve run drills or at least read through the spill and splash steps. Eye wash stations, safety showers, and spill kits need to stay clear and in working order. Make a habit of walking new team members through equipment every few months. Nobody plans on an accident, but habits and layout make all the difference when one strikes.
The more you work with chemicals like isobutanol, the easier it is to shrug off the warnings. Still, every splash, whiff, or spill shapes how you look at the lab the next day. A little respect for what sits in that bottle goes a long way toward a career with fewer scars—both the scary and the everyday kind.
Most people working with chemicals recognize that isobutanol and n-butanol both come from a family called butanols, but they often see just two things: industrial solvents and possible fuel sources. To many, the difference looks small—a switch in how the atoms are arranged. For anyone actually handling these substances or thinking about their environmental and safety impact, this tiny change leaves a big mark.
n-Butanol is a straight-chain alcohol, with all four of its carbon atoms connected in a single row. Isobutanol, sometimes called isobutyl alcohol, has a branch—three carbon atoms line up before the last one splits off to the side.
This shift in the structure means they act differently out in the world. n-Butanol has a stronger smell, evaporates less quickly, and mixes a bit better with water. Isobutanol, by comparison, tends to evaporate faster and feels less soluble in water, but it holds its ground in mixtures with gasoline and some plastics.
Working in labs and factories, I’ve seen how these small differences set the tone. In paint and coatings shops, n-butanol comes out as the preferred thinner for many products. Its slower evaporation reduces brush marks and leaves a more polished surface. Isobutanol works better in fuel blending, chosen for its similarity to gasoline and its way of cutting down engine knocking. Isobutanol even shows promise as a renewable biofuel. This isn’t just theory—petroleum engineers and lab techs measure real performance jumps on test engines using isobutanol, especially with stricter emissions standards around the corner.
The story goes beyond performance. n-Butanol’s strong odor and lower volatility make it more manageable in the workplace, but it still demands proper safety measures. Long exposure, even at low levels, can lead to headaches or dizziness. Isobutanol, on the other hand, calls for more careful ventilation since its vapors can catch fire faster and spread odor further.
Neither chemical is totally guilt-free in terms of environmental impact. Both break down pretty quickly in soil and water under normal conditions, yet spills can put plant and aquatic life at risk. Most seasoned workers treat both with respect, knowing that small leaks sometimes add up.
Companies often look for alternatives, and both types of butanol get compared to ethanol, which has a stronger record in fuel blending and lower health risks for short-term use. Yet, each butanol’s unique properties keep them important for certain jobs. Regulations keep pushing industries to reduce emissions and improve safety, which means manufacturers, scientists, and safety officers can’t just swap one alcohol for another without close examination.
Every week in the field, people highlight the importance of proper training, well-maintained ventilation, and strict storage practices. When handling isobutanol, keeping it away from open flames and hot surfaces makes a difference. With n-butanol, attention shifts to limiting skin contact and keeping containers tightly sealed.
The practical solution often involves blending the right chemistry with realistic safety measures. Companies turn to closed-loop systems and personal protective equipment because the risks remain real no matter how familiar these chemicals seem.
For those coming up in science or industry, it pays to look beyond the label. Understanding where n-butanol shines and where isobutanol pulls ahead leads to better decisions, safer workplaces, and less wasted time and money. It’s a reminder that even small changes in a molecule can shape jobs, safety plans, and the path toward cleaner technologies.
People might have never heard about isobutanol unless they work in a lab or spend time around fuel blends and paints. This colorless liquid has found its way into all kinds of products, from solvents to biofuels to cleaning agents. Anytime I read about another chemical being added to a household product or a fuel mix, I get curious about how safe it is for those nearby, breathing in fumes or accidentally coming into contact with it.
Isobutanol carries a distinct smell and evaporates more slowly than its cousin, n-butanol, but that doesn’t make it less of a concern. Workers in factories, labs, or even those using certain hobby supplies could run across it. So let's talk health: breathing in too much isobutanol vapors can bring headaches, dizziness, and nausea. In higher concentrations or during spills, it can knock people off their feet, both literally and figuratively.
Skin and eyes don’t do well with isobutanol, either. Splash some on your hand, and you’re in for redness, dryness, and potential chemical burns if you’re unlucky or don’t wash up quickly. Eyes react quickly—pain, watering, blurred vision. I’ve seen how poorly people recover from even a brief exposure to some solvents. Labs and workplaces emphasize eye and skin protection for good reason.
Short-term symptoms usually vanish once exposure stops, but longer-term effects stay less certain. Research hasn’t linked isobutanol to cancer in humans. Chronic lung or nervous system trouble doesn’t pop up much in studies, either. More information would help, especially since chemical exposure stacks over a lifetime.
No one should even think about drinking isobutanol. Swallowing it—on purpose or by accident—can hit the central nervous system hard and cause vomiting, confusion, and, in severe cases, depression of consciousness. There’s not much difference between it and other alcohols; they all cause damage inside the body, just at slightly different rates. I’ve been part of training sessions where antidotes and immediate treatment were stressed because even a small amount could harm adults and kids alike.
Spills can end up in soil or water, which isn’t just a paperwork problem for businesses. Isobutanol breaks down in the environment but can poison aquatic life if released in large amounts. There are international limits for how much isobutanol workers can breathe in over an eight-hour shift, so factories have to install ventilation and use chemical fume hoods. This isn’t just about meeting legal standards; it’s about protecting real people.
Practical fixes matter more than long warnings on a label. Proper training for everyone who handles isobutanol, even hobbyists at home, helps. Classrooms and labs need good airflow and ready access to washing stations. Clear labeling and storage rules make it harder to grab the wrong bottle or spill dangerous liquids. For workplaces, routine air monitoring and solid personal protective equipment can prevent most accidents. If health effects show up, early medical help beats internet searches every time.
Taking chemical safety seriously saves lives, plain and simple. Whether at a big manufacturing plant or a DIY garage, treating isobutanol with care makes a big difference in keeping people and the planet healthy.
Anybody who has worked around industrial chemicals knows you measure success by what doesn’t happen — leaks, fires, unplanned headaches. Isobutanol, a popular solvent and fuel additive, is no exception. Folks counting on it to keep business moving quickly learn shortcuts turn into emergencies fast. Storing and moving isobutanol safely isn’t only about following a checklist; it’s about thinking ahead, protecting people, and respecting what this alcohol can do if it escapes the tank.
Isobutanol burns. I remember a warehouse supervisor explaining, “It goes up quick if you get careless.” Its flash point is higher than ethanol or gasoline, but that doesn’t mean it won’t catch fire given the right conditions. Storing it in cool, well-ventilated spaces feels less like a suggestion and more like common sense to anyone who’s seen a spill go sideways. Even in small operations, using spark-proof tools and grounding drums stops static from turning into a disaster.
You can talk about vapor pressure and chemical stability, but real-life problems usually start with small spills or a drum stored too close to a heater. The National Fire Protection Association puts isobutanol in the flammable liquids category for a reason. It’s smart to use containers made of steel or certain polymers with solid seals. Most folks go for drums or ISO tanks with proper labeling, and I’ve never seen a wise crew skip regular checks for bulging, corrosion, or loose lids.
One piece folks learn the hard way: isobutanol loves to pull in water from the air. Once water sneaks in, it can mess with the quality, especially if the product is headed for fuel blends or industrial processes. Sealed drums and tanks cut down on this risk. Keeping an eye on temperature changes, which can lead to condensation, matters more than most realize until tanks start rusting inside or performance takes a hit.
Mixing accidents happen, too. Never store isobutanol next to strong oxidizers or acids. These combinations have a bad track record. I’ve heard stories of near misses, all from taking shortcuts on tank placement or skipping proper segregation.
Moving isobutanol between places had me anxious the first few times I watched a tanker roll out. It falls under hazmat rules for a reason. Department of Transportation regulations lay down clear steps, and honest operators double-check cargo certificates, tank integrity, and driver training before shipments leave the lot. It doesn’t take a major wreck to cause a problem, either — minor fender benders can become big news if containers rupture.
From my experience, communication counts as much as equipment. Folks receiving a delivery appreciate detailed handoffs and confirmation every container is sound and marked. Route planning and weather checks aren’t wasted time. Simple things — straps, proper tie-downs, secondary containment — have prevented more headaches than any fancy system I’ve ever seen.
Safe handling of isobutanol boils down to discipline. A shop, warehouse, or truck yard with clear rules and training creates a safety net that catches little things before they become headlines. It helps to treat every shipment as if it could go wrong — not because you expect it, but because the real world throws surprises fast.
People make storage and transport work, not just the containers or the rules. Paying attention, staying honest about small mistakes, and keeping the lines of communication open — these habits shield teams just as much as steel and plastic. That’s what keeps isobutanol a tool for progress, not a story on the evening news.