Talking about manganese carbonate means reaching deep into the story of progress. Early on, miners weren’t after manganese carbonate for its own sake but found it as a byproduct digging for other things. It didn’t sit in the limelight at first. Over time, people figured out that manganese carbonate could clean up chlorine gas and serve as a coloring agent for ceramics. Today, we see it playing a much broader part out in the world, shaping materials, steel, agriculture and beyond. Progress brought the shift from small-scale, curious use of natural minerals to the refining and chemical processes that bring enough manganese carbonate into the market to meet industrial need. Looking back, the path shows how old minerals find themselves at the center of new revolutions.
If you pull a sample from a supply warehouse, manganese carbonate usually presents as a pale pink to light brown powder. It doesn’t attract attention on first glance and could slip unnoticed past anyone unfamiliar. Yet this powder serves diverse industries, each with their own demands and standards. In agriculture, it’s considered an important micronutrient; in ceramics, a coloring agent; in the world of batteries and electronics, a stepping stone toward higher performance. The shape and purity of the material determine its suitability for a given use. The market sees both natural and synthetic forms, with the latter dominating where control of impurities matters.
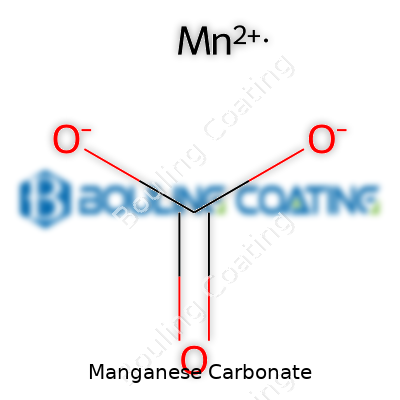
Chemically, manganese carbonate stands as MnCO3. Its color, often a modest pale pink, tells the trained eye something about the oxidation state and purity. The material won’t readily dissolve in water, but acids break it down to release manganese ions. With a density that puts it near 3.7 g/cm3, this compound won’t float, and its stability leaves it well-behaved under normal air and storage. Heat it strongly and it sheds carbon dioxide, forming manganese oxide—a property that finds use both in research and in industry. These traits stem from the manganese ion sitting at the heart of its crystal structure, locked in with carbonate groups.
Industry buyers look for specs like purity—often above 98% for high-end uses—particle size distribution, and moisture content. Impurities like iron or heavy metals draw scrutiny. The label has to warn about inhalation risks and proper handling, aligning with regulations where the product gets sold. Regulations on manganese carbonate have moved with the science; clear and detailed documentation shows respect for worker health and public safety. In labs and factories, those labels and data sheets spell out exactly what kind of material sits in the bag, reflecting a hard-earned understanding of risk and value.
In earlier days, natural mineral sources covered the limited demand, brought up from the ground and cleaned as needed. Increased demand led to development of chemical synthesis. One common route uses manganese sulfate and a carbonate source—like sodium carbonate or ammonium carbonate—reacted in water. Precipitation follows, and filtration, washing, and drying yield a reliable, relatively clean product. Researchers try to optimize this process for energy and water savings, reduce waste, and avoid dangerous byproducts. In some regions, producers still draw on natural rhodochrosite ores, but large industries depend on chemical production for consistent quality.
Manganese carbonate’s moderate reactivity makes it useful across chemistry labs. Expose it to strong acids; it breaks down to manganese salts, which fuel the next step in making manganese dioxide or other chemicals. Heating in air pushes it toward manganese oxide, useful in batteries and water treatment. Combining with a phosphating solution, manganese carbonate helps create conversion coatings on steel, putting a protective barrier against rust. Chemists keep finding routes to push its chemistry in new directions, making use of its ability to yield different oxidation states or react with other carbonates and oxidizers.
Sometimes, this compound shows up on paperwork as "manganous carbonate," nodding to its +2 oxidation state. Less often, the mineral name "rhodochrosite" refers to the natural crystal. Industry paperwork might list names in other languages, but the key identity remains the same—a modest yet reliable manganese source.
Anyone who handles manganese carbonate knows to respect its health impacts. Inhalation of dust stands out as the key risk, especially in facilities processing the powder in bulk. Chronic manganese exposure can lead to neurological effects over time—a fact that shaped occupational hygiene planning. Factories often use dust control systems, and workers rely on respiratory protection where dust can be generated. Local and international standards, including requirements for labeling, shipping, and handling, reflect decades of toxicology studies and hard lessons from experience. At the same time, safe storage keeps it away from strong acids to avoid unwanted reactions or exposure.
People reach for manganese carbonate across many fields. In agriculture, it feeds soils short on manganese, which crops need for enzyme function and growth. Crop yields improve when manganese found in carbonate form supplements poor soils, especially in intensive farming regions. Industry turns to manganese carbonate as a precursor for the battery-grade manganese oxides powering rechargeable cells found in everything from hospital devices to electric vehicles. Ceramics and glass manufacturers add this compound to produce warm pink, brown, or purple colors. Welders and metallurgists use it in specialty steel production. Water treatment plants sometimes use derived manganese oxides for purifying drinking water. Each application brings its own challenges and rewards, but all depend on steady supply and attention to quality.
Research around manganese carbonate reflects the shifting needs of society. A lot of focus lands on improving battery materials—making manganese-based electrodes cheaper and longer lasting without relying on cobalt or other expensive metals. Nanotechnology labs try to control the size and shape of manganese carbonate particles, hoping that smaller particles will increase reactivity or tune material properties. Soil scientists look at how its use might affect plant health beyond simple nutrition, considering trace contamination and how plants process different manganese sources. Ceramics experts keep chasing brighter, more stable colors and safer workplace conditions. Universities and institutes keep publishing on quiet questions—best practices for synthesis, dust exposure limits, all the way to life cycle assessment and environmental impact.
Decades of research anchor what we know about manganese toxicity. Acute exposure rarely kills, but long-term inhalation or massive misapplication creates risk. Manganese, essential at low doses, tips quickly into toxicity. Workers who spend years inhaling manganese dust can develop a neurological syndrome called manganism—one with symptoms that look a lot like Parkinson’s disease. Field studies in high-exposure farming regions push regulators to set lower application rates and better protective equipment standards. Ongoing toxicology projects test new forms and study cumulative exposure, recognizing that essential doesn’t always mean safe without limits. All this data keeps international agencies busy updating safety levels for food, water, and workplace air.
The next decades won’t let manganese carbonate drift into obscurity. Battery technology continues to grow, and the search for reliable, affordable materials becomes increasingly fierce. If researchers succeed at improving manganese-based battery chemistries, demand for pure manganese compounds will surge further. Green chemistry and environmental awareness pressure producers to cut waste and energy in making manganese carbonate, nudging industry toward more sustainable methods. Ongoing innovations in controlled-release fertilizers and ceramic pigments will call for new forms and purities, and safety research will keep tightening occupational exposure limits. Manganese carbonate, with its mix of steady tradition and untapped future, keeps reminding us how old chemicals find ever-new work to do as technology and society keep changing.
Manganese carbonate often flies under the radar, but its impact shows up in places we rarely think to look. On most farms, this mineral gives crops a solid shot at healthy growth. Plants don’t always get the nutrients they need from the soil alone, so adding manganese carbonate as a fertilizer keeps things on track. A wheat field can lose its yield if micronutrients like manganese run low, even when everything else goes right. Regions with sandy or overworked soil especially count on this supplement—it keeps the food chain moving and supports the farmers working that land.
This story extends into animal feed. Raising healthy livestock depends on more than just grain and hay. Diets for pigs, chickens, and cows often include manganese carbonate to support enzyme systems and proper bone development. Growth stalls and health problems follow when animals get shortchanged on minerals, but with careful mixing, these issues stay under control. Having visited family farms, I’ve seen feed blending up close—a few missing nutrients can throw the balance off for everything from milk yields to eggs produced.
Manganese carbonate’s work doesn’t stop at the farm gate. Source one of today’s lithium batteries and you’ll find its fingerprint there, too. Manganese compounds shape the structure and performance of battery cathodes, playing a key role in energy storage for electric vehicles and devices. There’s an environmental burden from mining and processing, but without these materials, scaling up clean transportation would slow to a crawl. Chemists have spent decades refining formulas that partly depend on manganese carbonate, and new projects keep tweaking these designs for more capacity and longer lifespans.
Move over to ceramics production, and you’ll see another slice of life that depends on this powder. Tile factories and potters use manganese carbonate for coloring glazes. The earth-tone browns found on kitchen tiles or bathroom fixtures owe their appearance to its steady hands. Not every pigment can stand up to firing in a kiln, but manganese carbonate rides it out without losing character. There’s also the glass industry, where it helps correct greenish tints and produce wine-clear bottles—an unsung trick that most people never notice.
Mining and refining any mineral raises concerns over worker safety and environmental care. Low exposure to manganese carbonate carries little risk, but heavy industrial use can raise dust and introduce complications. Workers handle the material using gloves and masks; regulators check sites for safe air quality. I once talked to technicians at a ceramics plant—they pointed out vents and dust traps, the kind of everyday technology that quietly keeps workplaces safe.
Looking ahead, the big question is how to stretch essential resources while cutting down on waste. Companies keep searching for alternative battery chemistries, yet right now, manganese carbonate stays locked into the supply chains. Agricultural advisors urge farmers to test their soils and only apply what’s needed. Even ceramics studios experiment with recycling methods that give used glazes a second life.
This mineral might not draw headlines, but by threading through everything from farmland to phone batteries, manganese carbonate proves that some unspectacular materials punch above their weight. As industries change, the challenge turns toward keeping its benefits alive without losing sight of environmental and health responsibilities.
Every time I dig into stories about chemicals, I think about how often workers and families get exposed to stuff they can’t even pronounce. This week the spotlight is on manganese carbonate. It’s a chalky pinkish powder, pretty cheap, and used in fertilizers, ceramics, and sometimes supplements. Seems harmless from the outside, but it pays to know what's really in play when people work with or live near the stuff.
Manganese itself is not new – it’s essential for health in tiny amounts. The trouble starts at higher doses or after repeated exposure. Manganese carbonate is no exception. Dust from this compound floating around a factory floor doesn’t just disappear. It settles in the nose and lungs, and from there, it can get into the bloodstream. Inhaling manganese dust for long stretches links strongly to mood problems, slowed movements, and something doctors call “manganism” – basically, a Parkinson’s-like brain disease affecting steelworkers and miners for over a century. Chronic exposure does its worst in jobs where little has changed since the hardhat and lunchpail era.
Are we talking instant danger? No, touching a bag usually won’t hurt, and it doesn’t blow up or catch fire easily. But safety gets dicey without proper controls. Fertilizer mixers, for instance, risk breathing in fine particles unless gear is up to snuff. I’ve known folks who complained about coughs and headaches after long shifts in poorly ventilated spaces, then later learned their companies hadn’t offered decent masks. It’s worse for those with existing lung problems: manganese carbonate can trigger asthma or make breathing harder over time. This plays out in stories from metal and ceramic factories, where workers sometimes chalk up fatigue or tremors to “a bad day” instead of the long grind of chemical exposure.
Rules do exist. The US Occupational Safety and Health Administration puts limits on dust in the air, but patchy enforcement is more common than you’d hope. Smaller outfits cut corners. Protective equipment disappears or isn’t replaced. Medical checkups become rare or superficial. I’ve seen supervisors encourage “toughing it out”, dismissing early symptoms as ordinary fatigue. No chemical is dangerous in isolation, but in a culture where shortcuts are tolerated, risks multiply fast. Manganese carbonate ends up being more hazardous than its dry, dusty look would suggest.
Addressing these problems doesn’t take moonshot technology. Companies do better when managers listen—when they invest in working hoods, make sure every bag-opening job happens with a mask, and encourage breaks to get away from dust clouds. Training shouldn’t just be a box-ticking chore on the first day; ongoing lessons and honest discussions about symptoms can catch problems early. Doctors need to include toxin exposure questions in regular health checks, especially for anyone who’s likely to carry dust home in their hair or on their boots.
On the larger scale, regulators and buyers have a part to play by demanding proof of responsible practices down the supply chain. No fertilizer or pigment should be cheap at the cost of safety. Personal experience and history show that protecting workers pays off in both loyalty and fewer sick days. Shielding communities and workers from harm calls for less shrugging and more straight talk about chemicals like manganese carbonate.
Science classes always tossed around chemical formulas and quirky names, but Manganese Carbonate stands out for more than the symbol MnCO3. On a lab bench, it doesn’t shout for attention. It usually takes the form of a light pink to pale rose powder, sometimes a soft, earthy tone that makes it easy to miss unless you know what you’re looking for. That color isn’t just for show; it tells a bit about what’s happening at the atomic level. The pink shade whispers clues about the manganese (II) ion inside, which guides a lot of what the compound sets out to do.
Manganese carbonate tends to avoid the headlines, but you’ll bump into its handiwork if you look closer at how batteries, ceramics, and even vitamins are made. People often forget how trace compounds like this power up some major industries. If you’ve ever taken a multivitamin, there’s a good chance you’ve swallowed a tiny bit of manganese, often starting off as this same pinkish powder before it changes form. The agriculture world puts it to work too. Crops depend on healthy soil, and sometimes a gentle touch of manganese carbonate helps get the nutrients flowing, fighting off plant diseases or patching up tired land.
On the flip side, large-scale use creates headaches when it drifts away from where it should be. Industrial mining and processing create dust and waste. Manganese itself isn’t rare, but careless handling has poisoned water sources before, leaving communities tangled up with pollution. Years ago, a mining incident in a developing country led to manganese salts washing into village streams. The outcome turned tragic for local kids. This wasn’t the fault of the chemical formula—it was people failing to plan for the consequences boats of powder and barrels of sludge can bring to real lives. Responsible management gets skipped too often because oversight takes effort and sometimes gets expensive.
Too often, solutions focus on technology or regulation, but then skip the follow-through. Plant operators should build in stronger controls—think double-checking waste handling steps, choosing closed processing loops, and not taking shortcuts with old equipment. Local governments need a seat at the table before anything gets dumped or poured. I’ve seen companies who rely on checklists and audits actually catch mistakes before they spill over into a big crisis. Community groups who get a transparent look at how minerals get mined or processed keep everyone more honest, and public reporting pushes companies to think twice before sneaking by loose practices.
We learn plenty about formulas, shapes, and reactions growing up, but rarely do schools connect those details to life beyond the classroom. There should be more space for questions about where these powders come from, where they end up, and who picks up the pieces if something goes wrong. Manganese carbonate might look gentle in a beaker, but like most elements, it takes people who care about its journey from mine to finished product to make sure it brings benefits, not just problems.
Walk into any warehouse handling minerals or chemicals, and there’s a good chance you’ll spot bags or drums labeled manganese carbonate. Its soft, pinkish powder creeps quietly into countless manufacturing processes, from ceramics to crop nutrition. The thing is, outside a tight group of insiders, few talk much about how to handle this stuff smartly. My own time around industrial supply rooms showed me pretty quickly that the basics make all the difference—yet they get skipped or rushed all too often.
Manganese carbonate doesn’t explode or fume. Still, it won't simply sit and wait for you patiently until you need it. It cakes with moisture and dusts up in the air, two traits that can muddy up the workplace and spark headaches for both workers and equipment. Store it somewhere dry, away from acids and strong oxidizers. Most folks do better by keeping it in sealed containers—think barrels with solid lids and labeled sacks stacked smartly, not slumped in cracked pallets or open bins.
High humidity or splashes from leaking pipes or spilled drinks really cause trouble. Wet or clumpy product clogs dosing heads and can corrode metal surfaces. Moisture also triggers slow chemical changes, which means the powder might not work as intended next time someone needs a precise blend. I’ve seen workers try to break apart solidified bags with makeshift tools, which rarely goes well and stirs up dusty clouds that no one wants to breathe.
This is where stories get personal. Even though manganese carbonate isn’t known for being brutally toxic, breathing in dust regularly is hard on the lungs, just like with most fine powders. Studies from industrial hygiene groups like NIOSH show a clear path from long-term airborne manganese exposure to neurological issues. No need for panic, but anyone handling it during weighing, pouring, or mixing really ought to have a simple dust mask at least. Goggles or glasses protect against accidents; all it takes is a wayward scooping motion, and suddenly someone’s squinting and rubbing their eyes, slowing the whole day down.
Walking through warehouses, I often spot forgotten piles of powder on the floor near mixing tanks or open bags dribbling along conveyor belts. Those spills don’t vanish—they build up and make the place feel sloppy. Sweeping with a regular broom isn’t ideal; it just kicks up more dust. Wet mopping picks it up more safely, and vacuum systems with HEPA filters work even better. Training staff to wipe up as they go and seal open sacks after use tightens up operations and keeps everyone safer.
Some rules work because they respect human nature: bins with lids at eye level, sturdy gloves in every size, labels with foolproof graphics rather than faded print. Posting reminders about safe handling helps, but walking a new worker through the storeroom, pointing at proper storage, and showing which scoop to use will last longer than another dusty manual. Integrating manganese carbonate storage routines into broader chemical management systems—complete with inventory checks—improves safety and saves money by cutting waste.
In my years on the shop floor and watching from the sidelines, the safest workplaces make the right way the easy way. For such a humble, useful compound, manganese carbonate deserves better than a forgotten pile in the corner or a busted sack under a leaky shelf. Safety isn’t magic; it’s just paying attention and building good habits, one day at a time.
I’ve stood in warehouses where bags of manganese carbonate wait for their trucks, and the story of packaging plays out on a practical level: it all starts with the material’s pinkish-tan powder, surprisingly fine, surprisingly dusty. I remember those early days learning about why packaging matters—not just to keep things neat, but to keep people and products safe.
Most manganese carbonate heads out the door in thick polyethylene-lined paper bags. Picture a bag that holds about 25 kilograms, sometimes as much as 50. These aren’t the flimsy ones from the grocery store. You’ll find triple-layered paper to add strength and, inside, a liner that keeps out the humidity. In the back of my mind, images of torn bags and fine dust clouds still linger, so hearing suppliers talk about moisture barriers rings true. Manganese carbonate reacts with air when it can, and wet clumping spells trouble fast.
Super sacks, or flexible intermediate bulk containers, show up when operations really get rolling. These hold up to a ton, can fill a forklift’s arms, and move quickly when time is short. Factories using a lot of material prefer these for their efficiency, but the same concerns about moisture and spillage apply. Occasionally, drums—typically plastic with strong sealing rings—show up in specialty orders, especially outside of bulk buyers. Again, it’s about protection: not just from moisture, but also from contamination or damage in transit.
One thing that stands out to me is how easily a packaging oversight can stop production lines or cause health hazards. A tiny puncture in a pallet’s bottom row, or humidity creeping through a seam, leads to caking and loss. Workers exposed to manganese carbonate dust face risks; respiratory irritation and long-term exposure carry worries that nobody working the line takes lightly. I’ve watched teams set up filtration fans and use gloves, but the frontline tool against exposure starts with a strong, sealed bag.
From time to time you see a supplier selling at a discount, but using single-layer bags or even recycled sacks. That’s when the risk of cross-contamination enters the conversation—especially for food or pharmaceutical sources. The lack of clear labeling or lot information makes tracking impossible if an issue arises down the supply chain.
So much of the industry uses the same old packaging because it works and it’s cheap, but the world’s asking for more. Workers want safer handling. Regulators push for traceability in every shipment. Food and animal feed producers demand materials that don’t pick up outside dust. It makes sense to see a gradual shift toward tamper-evident bags with QR tracing codes or even smart sensors that change color on moisture exposure. Recyclability lags, though. Most bags wind up in a landfill unless recovery programs are available.
Better packaging often costs a little more up front, but I’ve seen what happens when it saves thousands in lost material or downtime. Those who move toward stronger, safer options set a standard. If a manufacturer listens to feedback—say, a coating that resists moisture or extra-durable seams—workers and end-users both win. At the end of the day, packaging isn’t just a detail; it shapes safety, quality, and trust in every bag that rolls out the warehouse door.