Walk into any plastics manufacturing plant or look at the labeling on modern paints, and you’ll find traces of methacrylic acid. Chemists first documented this compound way back in the nineteenth century. In 1865, it came to light as a product described by C. D. Harries, showing up as a byproduct in organic synthesis. Since then, world wars, industrial revolutions, and the mass adoption of polymers have continued to push the frontiers of methacrylic acid. What started as a curiosity in early organic chemistry became a gamechanger in acrylics during the twentieth century. The invention of polymethyl methacrylate, known widely as Plexiglas or acrylic glass, is a direct offshoot and one that revolutionized aviation, construction, and automotive industries. From aircraft canopies in World War II to transparent barriers in banks and hockey rinks, methacrylic acid’s influence grows with every passing decade.
Methacrylic acid crops up in technical conversations wherever durable, weather-resistant plastics are mentioned. This chemical usually arrives as a colorless liquid with a piercing, sharp odor. Its boiling point lands at a modest 161°C and it hardens near 16°C, making it manageable for most operations. Unlike many organic acids, methacrylic acid mixes easily with water and many organic solvents. The double bond in its structure makes it highly reactive and essential for polymer science. In the lab, the acid stings the skin—literally and metaphorically—demanding respect and careful handling. Every time I open a new batch, that distinct smell reminds me how volatile and reactive small molecules can be.
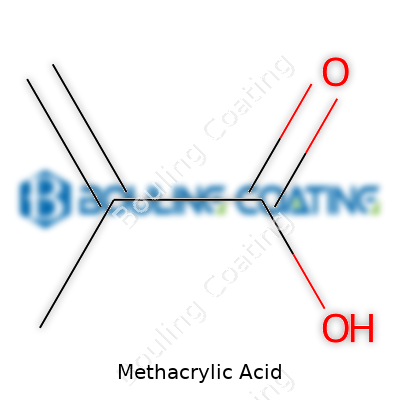
C4H6O2, molecular weight a hair over seventy grams per mole, with a melting point just above freezing—these numbers stick with anyone working with methacrylic acid. Hazard symbols mark every drum: corrosive, flammable, and environmentally hazardous, as regulated by most transportation authorities. Industrial users usually look for high purity, often above 99%. Labels often include synonyms like “2-methylpropenoic acid,” making it easy to miss that an unfamiliar product on a shelf is methacrylic acid in disguise. Brand names like Rohm’s “Methacryl” and the loose use of “MAA” by chemists keep the terminology just nuanced enough to confuse newcomers, especially if they’re not double-checking molecular formulas.
Producers today churn out methacrylic acid using a couple of dominant methods. The most established route starts with acetone cyanohydrin, reacting it with concentrated sulfuric acid to liberate methacrylamide, which then hydrolyzes to form the acid. This old process goes back decades but still finds use because of the robust supply chains backing it up. An alternative involves catalytic oxidation of isobutylene or tert-butanol, a process that takes advantage of better atom economy and sidesteps much of the hazardous effluent associated with cyanide chemistry. None of these routes are particularly green, and both demand vigilant controls to minimize releases of toxic byproducts.
Strong acid groups and a reactive double bond make methacrylic acid a chemist’s playground. Almost every major plastic and resin manufacturer relies on its ability to undergo rapid addition polymerizations. That’s how you wind up with PMMA—acrylic glass—or copolymers blended with acrylates and styrene for paints, adhesives, and coatings. Functionality doesn’t end with plastics. Its easily modified carboxylic group opens routes for new esters, which marketers dress up as “non-yellowing” or “high-clarity” monomers in the coatings world. The acid’s own reactivity, while an asset in manufacturing, means storage containers can become “hot”—building up heat if stabilizers aren’t present or if sunlight sneaks in through a warehouse window.
Whoever said chemistry had a naming problem wasn’t kidding. Methacrylic acid takes on titles like 2-methylpropenoic acid, alpha-methylacrylic acid, or just plain “MAA” on shipping manifests. In practice, the core structure doesn’t change, but brand distinctions sometimes promote modified versions, like hydroxyethyl methacrylate (HEMA) and butyl methacrylate, sold under specialty trade names. As someone who’s spent time untangling billing records, it always pays to double-check which “methacrylate” you’re actually receiving.
Methacrylic acid’s sharp smell is a built-in warning, but the real dangers cut deeper. Skin contact quickly leads to burns, and short-term inhalation causes eye and respiratory irritation. Emergency rooms sometimes see workers who underestimated flashpoints in poorly ventilated spaces. Strict workplace guidelines mandate splash goggles, chemical-resistant gloves, and plenty of airflow. Few forget the time an improperly capped drum started fuming in a warm loading dock. Regulatory bodies in Europe and North America put strict rules in place for storage, shipping, and personal protective gear, all based on hard lessons from past incidents.
Methacrylic acid’s reach stretches from dental fillings to weatherproof window panes. More than half the world’s supply flows straight into PMMA production, offering lightweight toughness in products ranging from eyeglass lenses to aircraft windshields. Water treatment plants rely on its copolymers to keep sludge from sticking and pipes running clear. Every time I squeeze out a tube of quick-dry adhesive to fix a broken lamp, odds are good methacrylic acid had a role in its chemistry. Older paints faded and cracked in sunlight before additives derived from methacrylic acid found widespread use. Medical devices also count on highly purified derivatives for contact lenses, bone cement, and tailored drug-delivery coatings, supporting advances that didn’t exist fifty years ago.
Every year, chemical engineers tackle the challenge of making methacrylic acid safely and cheaply. The scientific journals fill with studies on alternative feedstocks, including efforts to tap renewable biomass in place of fossil-derived chemicals. Academic groups dig into catalyst design to lower reaction temperatures or cut waste. Bio-based routes, like those starting from itaconic acid fermentation, catch plenty of attention, though high costs and scale-up hurdles slow their rollout. Cross-linked hydrogels built from methacrylic acid scaffolds now anchor entire sections of tissue engineering research, promising next-gen wound care and cell culture solutions. My own time in university labs showed how a seemingly “old” molecule keeps offering new chemistry.
Toxicity research put methacrylic acid under a harsh spotlight. Short-term exposure often leads to irritation, but repeated contact threatens to sensitize skin, setting off allergic reactions that can end laboratory careers. Animal studies show some inhalation risks and evidence points to potential organ toxicity at high doses. Though not classed as a broad-spectrum carcinogen, long-term chronic exposure brings enough uncertainty that regulations on emissions grow stricter every decade. Industrial plants work to limit wastewater releases, since aquatic organisms suffer at low concentrations. As countries strengthen chemical safety rules, companies keep searching for safer, less bioaccumulative alternatives, especially in consumer-facing products.
Methacrylic acid’s story feels nowhere near finished. Windows and dashboards made from its polymers keep getting lighter and clearer, and 3D printers now use photoreactive methacrylates for rapid prototyping. Startups experimenting with bio-fermentation keep hoping for a scalable, greener process, as fossil fuels grow scarce and climate rules tighten. Regulatory pressure and consumer awareness both climb, pushing companies to dial down toxicity at every stage, from plant emissions to final products. My own hope: someday, the “smell of chemistry” in a production plant won’t carry the sting that methacrylic acid brings today. Until then, this compound stays essential but double-edged, its future tied as much to safety and sustainability as to raw performance.
Most folks rarely hear the name methacrylic acid outside of a chemistry class, but chances are, it's already a familiar neighbor, quietly shaping daily experiences. As someone who worked summers in a plastics plant, I saw gallons of this clear, sharp-smelling liquid arrive by the drum, destined for all kinds of practical uses. At its core, methacrylic acid is a building block, a way for chemists and engineers to weld durability and clarity into so many essentials.
Start with one of the standout roles: acrylic plastics. Windows that stay clear, shower doors that don’t shatter as easily, and skylights letting in sunlight owe much of their strength to methacrylic acid. Technically, it makes polymethyl methacrylate, or PMMA, a tough and flexible plastic better known by brand names like Plexiglas. The world leaned hard on PMMA during the COVID-19 pandemic. Clear plastic barriers popped up in stores everywhere, silent protection built from methacrylic acid’s chemistry.
Teeth, too, get a direct benefit. Methacrylic acid transforms into dental resins, holding fillings in place and making dentures comfortable. Dentists trust it because it holds up against years of daily chewing. Step outside the clinic and this acid lives on in paints and coatings, keeping colors bright and surfaces smooth. Car makers, architects, and artists all reach for methacrylic acid-based polymers when they want results to last and look sharp.
Paints need more than color; they need to stick, dry, and resist the weather. Methacrylic acid makes modern coatings better at clinging to wood, metal, and concrete. It helps stop rust, chipping, and fading, which means fewer repairs and less waste over time. In my hometown, city workers paint playground gear with coatings derived from this sturdy acid. Even after harsh winters and wet summers, those slides and swings still shine.
Textiles also rely on this valuable chemical. Clothes that stretch and bounce back, athletic gear that doesn’t sag, and fabrics holding bright prints—all get a boost from methacrylic acid in synthetic fibers. This brings comfort and style into closets without needing frequent replacement.
No commentary on methacrylic acid feels complete without addressing what happens after people enjoy the benefits. Factories producing ethyl and methyl methacrylate from methacrylic acid generate emissions that communities often worry about. Methacrylic acid is not the most dangerous substance out there, but it can irritate skin and eyes, and breathing it over time in factories demands serious safety steps. Regulators and manufacturers already aim for tight controls. Moving toward greener alternatives, reusing waste, and keeping air and water safe will take more investment, but everyday folks and workers both stand to gain in the long run.
Leaning on methacrylic acid lets innovations stick, shine, and stay strong. With smart choices and better stewardship, society can hold on to its benefits while protecting people and the world around us.
Methacrylic acid doesn’t have an easy name, but its impact is pretty direct. As someone who’s seen workshops, labs, and small factories up close, seeing a bottle of this stuff on a bench always sets off a few mental alarms. There’s good reason for that. Methacrylic acid, used in the plastics and coatings industries, can hit hard if it’s handled without care.
Inhaling methacrylic acid vapors makes it hard to breathe. It can sting the nose and lungs, making your chest feel tight. Skin contact can cause burns, redness, or pain. Eyes feel the worst of it, where a splash can bring on immediate watering and damage. It isn’t rare to see cases in medical journals where accidental exposure sent workers for treatment. Breathing protection and gloves aren’t overkill in any workshop using this acid.
This chemical still has a place in the industry. It helps make flexible plastics, dental materials, and paints. The problem crops up with sloppy short-cuts. Rushed workers, poor labeling, and corners cut for cost savings magnify risk especially on small sites or in developing countries. One incident I learned of in a paints facility caused serious burns to a technician’s hands, all from grabbing a leaking bottle without gloves.
Mistakes ripple beyond personal health. Spills end up in wastewater. In places without proper disposal, that water can contaminate soil or pollute rivers. Methacrylic acid doesn’t break down quickly in the environment; fish and plants suffer if exposure happens regularly. Regulatory agencies in the U.S., Europe, and Japan—where environmental rules have teeth—list this chemical as hazardous for a reason.
Accidents expose gaps in both training and oversight. Safety data sits in manuals, but does anyone bother to read more than the bolded warnings on a dusty folder? Visiting sites and asking workers reveals a pattern: real-world practice doesn’t always match policy. People skip goggles for comfort, or new hires get just a brief safety rundown rather than hands-on demos. Not until someone gets a scare do habits change.
The hard part comes in holding both companies and individuals accountable. Company leaders control training, invest in the right protective gear, and push for automation in dangerous steps. But they also face a real temptation to do things quicker and cheaper, especially in highly competitive markets. On the worker side, diligence sometimes slips in the rush to hit output targets. What changes things is a real culture of safety—not just paperwork, but walk-throughs, drills, and visible enforcement.
Better engineering helps, like sealed process lines and automatic dosing, so hands don’t get near raw acid. Regular air monitoring can flag vapor leaks before they become an emergency. Medical follow-up matters for workers exposed over years. On the regulatory end, stricter tracking of waste and higher fines for infractions send a message. Open access to education, especially for contractors or workers in smaller operations, can narrow the knowledge gap that leads to accidents in the first place.
Methacrylic acid isn’t going anywhere soon. The risks aren’t a mystery, and real leadership means keeping those risks from quietly building into disasters. Every splash and inhalation offers a lesson—one no workplace should have to keep learning the hard way.
The chemical formula of methacrylic acid is C4H6O2. That string of letters and numbers may not mean much for most people, but there's a whole world of impact built into those few elements. Every time methacrylic acid comes up, most folks in science and manufacturing circles see the building blocks for familiar things—acrylic sheets, all sorts of plastics, coatings, adhesives, paints, and even some dental materials. Dig a little deeper and it’s easy to spot ways this one compound shapes entire industries and reaches into our everyday lives, often unnoticed.
In practice, methacrylic acid shows up just about everywhere—in those clear acrylic barriers at checkout counters, weatherproof paints, lightweight panels, and medical devices. The reason: it brings both strength and flexibility. Factories across the globe mix methacrylic acid with other chemicals, sparking reactions that lead to durable and tough polymers. These are what make certain plastics resistant to weather, chemicals, or shattering. Take a look around a hospital, dental clinic, or even a construction site, and there's a good chance something traces back to this single chemical compound.
Sometimes people overlook how the simple layout of C4H6O2 connects right back to stubborn stains that wash off easily, surfaces that stay clean for years, and lightweight gear that handles serious wear and tear. Industries keep reaching for methacrylic acid because alternative chemicals often can’t deliver the same balance between toughness and workability.
Like a lot of industrial chemicals, methacrylic acid comes with a safety story that’s not always front and center. On its own, it has a sharp smell and can be rough on the skin, eyes, or lungs. Workers near the stuff count on gloves, goggles, and well-ventilated spaces to cut the risk. Environmental agencies have a close watch on how it’s used and disposed of, because leaks and spills could end up harming water supplies or wildlife downstream.
Decades ago, people learned the hard way that not every chemical designed for industry belongs in every river, backyard, or workspace. So regulations grew around methacrylic acid’s production and shipping. Most countries keep things tight to avoid accidents, but there’s never a full stop—demand for plastics and related materials shows no signs of dropping, and this chemical remains in heavy rotation. Tackling safety means supporting research into alternative compounds, improving extraction techniques, and making sure the people handling these chemicals every day have better training and protective gear.
Given its deep roots in so many products, methacrylic acid won’t disappear overnight. Instead of aiming for replacements and greenwashing, a more practical route sees industries investing in smarter controls, keeping waste to a minimum, and making recycling routines more robust. Researchers who dig into this chemistry often focus on refining catalysts and tweaking processes so that every batch has less waste, pulling the industry closer to a safer and more sustainable setup.
Learning about the formula—just four carbons, six hydrogens, and two oxygens—acts as a reminder: chemistry on the page matters mostly because of how those molecules shape the world around us. The focus on both innovation and responsibility will keep methacrylic acid useful without letting safety or the environment take a backseat. To keep moving forward, honest conversations and clear-eyed science need a stronger presence than shortcuts.
Most folks outside of lab work rarely cross paths with methacrylic acid. Once you get involved in manufacturing or chemistry, handling such materials starts to feel routine. Complacency creeps in if you aren’t careful. Methacrylic acid rewards that attitude with accidents. This stuff can burn your skin, make your eyes water, and its vapors do worse in a closed room. Protecting people starts where you store the drum—never just in some corner with paint cans.
Methacrylic acid demands a cool, dry storage spot. Heat means trouble. At higher temperatures, this chemical builds pressure and risks polymerization. I've seen barrels warp with improper storage, fume hoods overwhelmed, alarms blaring because someone stacked containers near a boiler room. That situation can force a plant shutdown.
Tight seals on containers save you from unnecessary headaches. Moisture creeps in through a broken lid or rusted metal. Water can trigger an exothermic reaction that turns a leaking drum into a toxic mess within hours. In my experience, even experienced teams underestimate just how corrosive a few drops of water can be alongside the acid.
Chemicals like methacrylic acid join other organics in wanting their own labeled spot, away from oxidizers and too much sun. I’ve seen good people trust cardboard labels instead of the large print on the side of original drums. Use durable tags—otherwise, nobody’s sure what’s what after a few months and faded ink. The cost of a new tag is nothing compared to a misidentified spill.
Ventilation truly separates safe storage from risky corners. A closed room without airflow traps vapors. Methacrylic acid vapor isn’t just foul—it builds up and eats into electronics, wiring, or your lungs. Exhaust fans and open racks stop that cycle. Upright barrels cut leaks. Spacing keeps containers from bumping and breaking.
Staff turnover breaks good habits faster than most realize. New hires, eager to please, stack containers too close out of ignorance or because space is tight. Repeated hands-on training fixes that more often than a fresh manual. Walk through the storage area. Point out old spills and scars on the floor. Share why these things matter, not just that they do.
Methacrylic acid isn’t going away as long as plastics and polymers stay in demand. Investing in spill pans, chemical-resistant racks, and alarming for temperature swings sounds expensive, but doesn’t come close to the cost of hazmat response or lawsuits after poor handling. I’ve seen facilities start with one spill and end up with audits, fines, and mandatory upgrades. The money saved by skipping safety now turns into a much bigger bill down the road.
Good storage affects more than just those on the warehouse floor. Fumes seeping into shared ventilation can drift into offices next door. Sprinklers hitting a leaking drum turn a warehouse problem into a neighborhood scare. Local fire crews rely on clear, honest labeling to know what’s inside before a spill or fire.
Respect for chemicals is a learned behavior. Methacrylic acid doesn’t forgive laziness. Small steps like regular audits, proper signage, and a culture of double-checking bring everyday protection—not just compliance, but real safety for everyone breathing the same air.
Look around, and you’ll spot a footprint of methacrylic acid in places most folks never suspect. I’ve watched it change the way people drive, care for their homes, and even treat their teeth. This compound isn’t a headliner, but it deserves credit for making a lot of things work better or last longer.
Few things bug drivers like scratched or foggy windshields. The car industry leans on methacrylic acid because it helps create durable poly(methyl methacrylate) (PMMA), often known as acrylic glass. You see it in headlights and taillights too. There’s value in using a lightweight material that holds up against cracks, weather, and sunlight much better than old-style glass. Every time I notice a clear, shiny headlight after years on the road, I remember how material science, not just design, keeps us safe and improves visibility at night.
Anybody who has watched paint peel off a shed knows the cost of inferior coatings. Some of the toughest, most weather-resistant coatings use methacrylic acid-based polymers. Coatings with this chemistry offer better adherence to metal or wood, brighter color that resists fading, and surfaces that clean up more easily. For folks in the construction or renovation business, these qualities save time and money by cutting back on repairs and repaints. These improvements don’t just mean better looks—they translate to longer-lasting infrastructure and products.
From consumer electronics to sturdy construction gear, strong adhesives keep products from coming apart under pressure. Methacrylic acid steps up by making adhesives that stick better to tricky materials, including plastics or metals that tend to resist glue. I’ve handled plenty of products where a smartly designed adhesive line made all the difference between a product lasting years, or splitting apart after a short drop.
Few people think of chemistry when they sit in the dentist’s chair. Methacrylic acid forms the base for materials used in dental implants, fillings, and dentures. Dentists trust these materials for their clarity, their strength, and their gentle feel in the mouth. Patients benefit, too—better dental resins lead to longer-lasting fillings that fit more comfortably. Hospitals also use methacrylic-based plastics for medical devices, where clarity, safety, and stability count for a lot.
Textile makers and hygiene product manufacturers rely on polymers derived from methacrylic acid to create superabsorbent fibers and fabrics. Think diapers that stay drier, or activewear that wicks sweat and withstands repeated use. From what I’ve seen, the link to basic comfort in daily life comes straight from advances in specialty chemicals like this one.
Use of methacrylic acid brings up tough questions about waste, recyclability, and workplace safety. Traditional production routes still pull from fossil fuels and can create environmental byproducts. As more industries call out for greener solutions, researchers and companies look for new ways to make this acid from plant-based materials or recycle methacrylate plastics more efficiently. Supporting these shifts could mean less pollution and better resource use. For consumers, demanding clear labeling and sustainability disclosures can push the market toward safer, cleaner options without losing out on product quality or performance.