There’s something fascinating about the way chemistry shapes the world, and methyl methacrylate stands as a prime example. Its history starts back in the 1930s, when researchers began to look for alternatives to glass, driven by both curiosity and need. Ludwig Gattermann’s students in Germany played a major role in piecing together its structure, but it took major chemical companies, particularly in Europe and North America, to recognize its massive industrial potential. What began as a laboratory curiosity soon found commercial value in wartime, as polymethyl methacrylate (PMMA) replaced glass in airplane canopies and submarine periscopes. This switch saved countless lives, since PMMA shatters without sending shards flying, unlike its glass rival. Over the decades, methyl methacrylate slid seamlessly into everyday life—signage, automotive parts, medical devices, even dental work. Its rise wasn’t about chasing a chemical novelty; it was about filling real needs, safer products, and better manufacturing options.
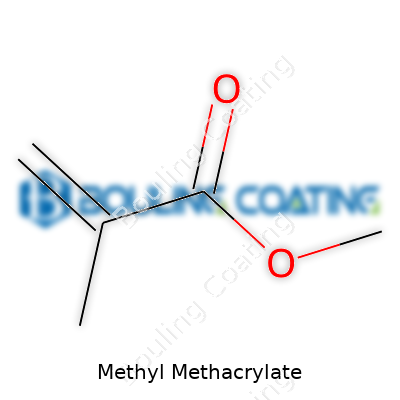
Dive into any supply of this clear, colorless liquid, and some truths become clear quickly. Methyl methacrylate carries a distinctive, pungent smell. It doesn’t play nicely with water, preferring its own company much like oil and water arguing in a bottle. This chemical turns to vapor at room temperature, so inhalation risk has led to careful handling rules. It boils at a temperature just above 100 degrees Celsius and solidifies in chilly storage. Its molecules feature a double-bonded carbon, making it ready for change—this fuels quick polymerization and underpins its value in plastics. What surprises many is the way it cuts a wide path through industry: its relatively low viscosity lets it slip easily into molds and casting processes, giving manufacturers plenty of design freedom. Its chemical formula, C5H8O2, means its backbone isn’t complicated, but that simplicity invites a world of adaptations.
Getting methyl methacrylate isn’t a simple affair, even though it looks straightforward on paper. Industrial plants usually rely on the acetone cyanohydrin process, which involves reacting acetone with hydrogen cyanide, then hydrolyzing the product with sulfuric acid and methanol. This approach draws from economics as much as chemistry—available starting materials, energy use, and waste control all shape industry choices. Over time, newer catalytic processes have started to replace the old cyanohydrin cycle, mainly because they produce less hazardous waste. Meeting technical standards, set by groups like ASTM and ISO, isn’t optional—manufacturers keep close tabs on purity, inhibitor levels, and water content, since impurities trigger runaway reactions or dangerous byproducts. Storage calls for outgassing inhibitors and well-ventilated tanks, since the monomer can polymerize explosively without warning. Labeling hinges on clear hazard markings, warnings about volatility, and batch tracking to guarantee safety and traceability all the way to the consumer.
Methyl methacrylate doesn’t really sit still. Its carbon double bond invites attack by free radicals and other initiators, turning this small molecule into massive chains or crosslinked networks. This reaction isn’t just a laboratory trick; it’s the core of PMMA production and why acrylic plastics can be tailored for clarity, toughness, or flexibility. Because the double bond is so reactive, chemists attach side groups—esters, acids, or other functional arms—to tweak pliability or color. This sort of modification lies behind the glow of rear car lights, strong display cases, or even high-performance adhesives. Working with methyl methacrylate, anyone in chemical manufacturing needs to respect its tendency to run wild in heat or sunlight—unstable mixtures can harden in pipes and tanks if not handled with caution. But with the right know-how, this same wild streak becomes the foundation for paints, coatings, and ultra-transparent barriers that outperform glass.
Walk into a factory, dental lab, or hardware store, and the same chemical gets called by dozens of names. Methyl methacrylate goes by MMA on drum labels and safety sheets. In the trade, PMMA means the big brother polymer, also sold as Plexiglas, Lucite, or Perspex, depending where you live. The names change across borders, but the chemical behind the label hasn’t shifted. These names matter because supply chains and regulatory filings use them interchangeably, and workers need to know what’s in their hands. Safety standards also refer to this alphabet soup, making it vital to cut through confusion. The industry language might trip up outsiders, but for those who shape plastics, clarity on naming saves money, time, and frustration.
Experience in manufacturing and lab work shows that methyl methacrylate is nothing to take lightly. Like other volatile organics, it evaporates quickly, creating inhalation risks. Short exposures cause irritation to the eyes, skin, and respiratory system. Extended exposure or poor ventilation heightens these effects. There have been incidents where improper storage led to explosions—MMA can polymerize inside closed drums, generating enough pressure to burst them. That risk motivated strict rules: keep inhibitors in every batch, track storage temperatures, and use explosion-proof gear for every pump or tank. Fire departments treat MMA as a high flammability risk. Chemical plants install leak detectors, use fire-resistant building materials, and run regular training sessions. If an accident does happen, emergency protocols swing into action because the risk extends fast in confined spaces. I’ve spent hours reviewing incident reports, and most stemmed from cut corners on ventilation or skipped inhibitor top-ups. No product is worth a life; safety rules grow out of real-world lessons, often learned the hard way.
Open your medicine cabinet, walk into a hospital, jump in a car—chances are strong you’re bumping into methyl methacrylate or its polymers. In medical fields, MMA transformed bone cements and dental implants, prized for remaining stable inside the body and low allergenic profiles. Its use in making tough acrylic glass isn’t just about clarity, it’s about keeping people safe, since shattered PMMA rarely injures like traditional glass shards. Road signs, skylights, aquariums, protective shields in sports arenas—all count on the toughness and light transmission that MMA brings. Artists and manufacturers alike favor its flow in paint and coating formulations, since it keeps colors bright and surfaces smooth. Even nails in salons owe their long-lasting shine and sculpted look to MMA’s versatile chemistry. Whenever engineers and product designers push for sleek, safe, and durable finishes, this chemical sits behind the scenes making it possible.
The story of methyl methacrylate didn’t stop at its first boom. Academic labs and corporate R&D teams push for greener ways to make and recycle MMA, seeing mounting pressure from environmental groups and stricter regulations on hazardous waste. Biobased routes, using sugars or agricultural waste, promise smaller carbon footprints and fewer toxic byproducts. Meanwhile, improved catalysts cut down on waste acids and energy used per kilogram produced. These advances matter because global demand for lightweight plastics keeps rising, hand in hand with demand for better recycling systems. Research into “closed-loop” recycling of PMMA could turn post-consumer plastics back into fresh MMA, easing pressure on both raw material supply and landfill use. Developing additives that boost UV resistance or lower fire risk keeps research labs busy, always aiming for safer, longer-lasting products. Ongoing research into the environmental and health effects of MMA, particularly microplastics derived from its polymers, pushes the field forward and sets the standards for tomorrow’s uses.
Despite its many uses, concerns about methyl methacrylate’s toxicity linger. Short-term inhalation brings headaches, nausea, and eye irritation in some people. Most research points to low long-term toxicity at typical exposure levels, but certain animal studies suggest repeat or high doses could impact organ function. Regulatory agencies like OSHA and the EPA set clear workplace limits, and industry groups share extensive safety data. The skin allergy risk, particularly among dental technicians and nail salon staff, sticks out as a recurring issue—I’ve heard of workers developing chronic dermatitis from repeated, unprotected contact. Companies responded by upgrading ventilation, gloves, and education. As for fears about cancer or birth defects, the weight of current evidence shows little risk at usual exposure, though scientists remain vigilant. Personal experience in workshops brings a simple lesson: respect the handling instructions, know your limits, and recognize early symptoms before they grow. Government safety rules and transparent sharing of data keep public health risks in check, but individual responsibility still counts for a lot.
Rising pressure for sustainable solutions calls for a big rethink in MMA production. Green chemistry approaches—catalytic routes, recycled inputs, lower emissions—are moving from concept to factory floor. As plastics become a political issue and public scrutiny zooms in on microplastics, MMA producers invest in safer, more recyclable blends. The push for transparent, shatterproof materials sets off new uses in electronics, renewable energy, and public transit, not to mention art and architecture. As digital manufacturing spreads, 3D printing ties into MMA chemistry, opening up direct printing of optical parts and custom implants. I see change already—young researchers blend traditional strengths with fresh thinking, challenging old models for waste, safety, and design. Health, climate, and raw material limits force hard choices, but MMA’s combination of clarity, toughness, and chemical adaptability gives it staying power. The next decade holds a wild mix of market shifts, regulatory change, and technical leaps, but I’d bet on methyl methacrylate keeping its place in the spotlight. Its future won’t repeat the past, but it draws from everything learned so far—lessons in chemistry, industry responsibility, and real-world impact.
Step into a hospital, flip through a glossy magazine, or roll across a fresh stretch of highway—you’ve brushed up against methyl methacrylate in ways you likely never realized. On the surface, this chemical has a name fit for a tongue twister, but beneath all those syllables, it shapes more of our modern world than gets talked about at dinner tables.
Methyl methacrylate, often known by its initials MMA, drives the creation of one of the most recognizable plastics: acrylic, or polymethyl methacrylate. Acrylic sheets often go by names like Plexiglas or Lucite. They pop up wherever glass feels too risky or heavy, like in airplane windows, aquariums, or protective barriers in hockey rinks. These plastics don’t shatter like ordinary glass, and they weigh quite a bit less, so more public spaces have traded the old for the new.
Medical staff count on MMA-based bone cement to anchor artificial joints inside the body. Think of hip and knee replacements—areas where weak adhesives could mean pain from loose implants, infections, and all kinds of trouble after surgery. Surgeons want something reliable, and MMA-based cement brings both strength and enough versatility to support a growing aging population. That real-world value goes deeper than just numbers; more reliable recovery means people get back on their feet and into their lives faster.
Road engineers paint crosswalks, bike lanes, and highway lines with MMA-based paints for a reason — they don’t just stick, they last through storms and sunshine, car tires, and heavy trucks. Safety on the roads often hinges on how well drivers see lines at night under headlights or when it starts to pour. MMA’s durability keeps lines bright, which gives everyone a better shot at making it home safe.
Artists and builders love acrylic resins, not just for clear panes but for museum displays, light installations, even complicated sculptures. Shattering a glass display case during an opening night party makes for a rough memory; acrylic steps in for these moments, keeping things sturdy without hiding the view.
Breathing in MMA fumes at work can make people sick with headaches or nausea, and some studies point to lung or skin issues among exposed workers over time. I’ve heard from industrial workers past and present about the importance of strict ventilation and good training. People deserve protection, plain and simple. Plants need to actively monitor air quality, offer protective gear, and provide rapid support for exposed workers. Public pressure for worker safety changed asbestos practices decades back, so the same level of care should apply here as well.
Right now, most MMA production comes from fossil fuels. That worries environmental groups, and it gives manufacturers a reason to look at greener routes—including processes based on renewable materials. Some pilot projects try using plant-based sources or recycling old plastic into new MMA. These aren’t sweeping the market yet, but early steps toward renewable production matter for cutting long-term emissions.
Methyl methacrylate shows up in countless spaces, helping people live, heal, and travel more safely, but it doesn’t come without headaches—both literal and figurative. Smarter safety rules, tougher air controls in plants, and real investments in greener chemistry all help smooth the rough edges. As more eyes open to the everyday uses of MMA, public demand can push for cleaner and safer innovation, keeping this everyday chemical part of the solution rather than a hidden problem.
Factories smell a certain way, especially when plastic parts start to shape up on the production line. That sharp, almost sweet odor? Often, that’s methyl methacrylate. People don’t think much about chemicals in daily life, yet this substance plays a big role in dental work, prosthetics, Plexiglas, flooring, and automotive parts. Few outside the manufacturing loop even notice the word “methyl methacrylate.” In many shops, folks have felt it in their noses and throats before they ever saw it in print.
People handling methyl methacrylate day after day understand what it does to the body. Some catch a headache or a cough after only a couple hours surrounded by its fumes. Eyes start to sting and sinuses rebel. The body gives clues long before technical data sheets get passed around. NIOSH and OSHA have weighed in because workers have reported rashes, eye burns, and tightness in the chest. A day’s exposure might send some home coughing, with hands turning red and cracked from spills.
A coworker once worked in a lab where methyl methacrylate was poured in big batches. She’d say, “If you smell it, you’ve been exposed.” The labs had fancy vents. Still, some days the whole floor reeked of it—ventilation couldn’t keep up. Later, that coworker developed a nagging cough and lingering skin irritation. Doctors listened and nodded, but warnings only helped so much if the pace picked up and safety steps seemed like they slowed everyone down.
Research doesn’t hedge much—methyl methacrylate works well as a quick-reacting monomer, but exposure causes definite problems. NIOSH has listed it as a respiratory irritant and recommends limits in air to bring down risks of throat and lung trouble. In some cases, people working years with this chemical have developed occupational asthma. Medical journals report skin contact leading to chemical burns and dermatitis. What once seemed like a minor work nuisance often turns out to mark the start of something chronic.
Years ago, nobody wore gloves mixing and pouring. Now, data from the U.S. Environmental Protection Agency and similar organizations in other countries tell everyone—protect the skin, guard the air. Some folks still think a bad smell is the only thing to worry about. It’s more than that: allergic reactions add up over time. Once someone gets sensitized, even tiny traces in the air can trigger asthma attacks.
Solutions tend to start with better information and honest conversations at work. Good ventilation is not just a box on a checklist; it saves lungs and keeps people out of the hospital. Gloves and goggles actually make a difference, and reminders from safety crews help, especially for new hires who don’t know any better than to wipe a chemical off with their bare hand. Supervisors who listen to complaints about smells and rashes set a standard that counts for something.
Switching chemicals is tough, so workers and managers need strong policies that make protection easy, not an afterthought. Simple regular breaks outside the work area, plain reminders before a shift, and access to washing stations help more than the thickest safety poster. Companies that track health complaints and air levels spot patterns before injuries become lawsuits—or worse, permanent health problems.
Methyl methacrylate keeps parts moving and products cheap. Still, its toll on human health needs attention at every step, not once a year at a safety refresher. People have the right to come home safe, not just get by until payday. Risk is part of the job—so are the solutions that put workers first.
Walk into any facility that handles methyl methacrylate, and you'll sense a mix of urgency and routine. This isn't your average chemical. With its flammable fumes and unpredictable behavior, storing it asks for more than just metal shelving and a warning sign on the door. Too many news headlines have highlighted the havoc one mistake with methyl methacrylate can trigger—from warehouse fires to environmental nightmares.
Methyl methacrylate is famous in the plastics world, but notorious for being volatile. Just a small spark around an open drum can produce a disaster nobody wants to clean up. Unlike water or canned goods, storing this liquid takes a real commitment to safety. It prefers cool, well-ventilated spots, kept far from heat, open flames, and incompatible chemicals like oxidizers. I've seen operators cut corners, thinking a nearby electrical panel wouldn't hurt. Accidents don’t care if somebody means well—chemicals like this demand respect every day.
Temperature swings also spell trouble. In summer, storage tanks heat up and pressure builds, so without relief valves or temperature alarms, something's bound to go wrong. There’s no substitute for regular checks on tank integrity. Leaks can go unnoticed until there’s an overpowering odor or neighbor complaints. At that point, the situation can snowball fast.
From personal experience, I know the headache after breathing in the fumes, even at low levels. Not everyone realizes methyl methacrylate vapor hangs in the air and irritates more than just the nose. Good ventilation does more than help the air feel fresh—it keeps the workplace out of the emergency room.
Adequate labeling helps in the best way—no worker should mistake this for a harmless solvent. Signage, colored barrels, and dated inspection stickers aren’t just regulatory hoop-jumping. These save lives. Many small businesses, unfortunately, don’t make this investment and end up training new employees on the fly, sometimes with tragic results.
While regulations like OSHA’s flammable liquids rules offer some guardrails, real peace of mind comes from a team that practices safety instinctively. Training sessions, fire drills, and reviewing near-misses turn nervousness into muscle memory. Installing explosion-proof lighting and grounding all containers often sounds costly until you see the aftermath of a fire.
I’ve noticed many storage mistakes boil down to weak planning and tight budgets. Straightforward solutions—like keeping supplies in original containers with secure lids, keeping storage areas separate from production, and managing inventory to avoid overstock—drive meaningful change. Smart operators build strong ties with local fire departments and run through emergency scenarios. This builds the kind of confidence that checklists alone can’t provide.
The storage choices for methyl methacrylate stretch far beyond regulatory checkboxes. Safe storage reflects an organization’s culture and its care for workers and neighbors. The companies that get this right show up in the community as trustworthy partners, not ticking time bombs. Real expertise isn’t about memorizing every number in a rulebook; it’s about building a culture where people prioritize each other’s safety with every drum stored and transferred. I’ve seen the difference. It’s real, and in this business, it’s worth the work.
Methyl methacrylate pops up anywhere from dental offices to industrial plants. Its sharp, almost fruity smell hangs in the air, a telltale sign that careful steps matter. Plenty of folks view the stuff as just another chemical, but that attitude can cause big trouble. Anyone working with methyl methacrylate has a responsibility—both to themselves and coworkers–to take safety seriously, not out of fear, but out of respect for real risks.
Methyl methacrylate comes with clear risks. Short contact with the skin leaves redness or irritation. Breathing in its vapors stings your eyes and rattles your airways. People who work in spaces filled with this chemical often develop sensitivities that never fully go away. There have even been cases of asthma that linger after folks switch jobs. Some sources even point to memory issues among workers exposed for long stretches. These aren’t just rare horror stories. They’re the sort of warnings anyone in the field hears sooner or later.
It only takes one bad spill or a moment of inattention to trigger headaches, dizziness, or even vomiting. Several fire departments flag methyl methacrylate as especially flammable, and it doesn’t need much to catch—from a heat lamp to a tiny spark on a static-charged jumper. It spreads quickly along surfaces if released, leaving both injury and property damage in its wake.
Good habits are worth more than fancy labels. I’ve seen rooms with top-notch fume hoods and nobody bothers turning them on. Complacency grows fast in familiar settings. The best safeguard? Treat every day like training day: gloves, goggles, and old-fashioned ventilation stand front and center. Keeping container lids tight, checking for leaks each morning, and never skipping protective gear isn't paranoia–it’s just habit.
Spills invite disaster. Simpler routines tend to stick. Even a layer of absorbent pads near mixing stations makes a difference. Regular checks on spill kits show the team cares about safety, not just compliance. Talking through what to do during a spill, or running drills a few times a year, builds muscle memory. If folks see cleanup as teamwork, there’s less chance anyone freezes up during a real emergency.
Rules exist for a reason. Agencies like OSHA and NIOSH don’t just write boring manuals; they save lives. Limits on exposure in the workplace don’t ask much—just a little planning. Rotating work shifts or rotating tasks gives staff a break from fumes. Posting hazard signs lets new hires know what’s at stake without making anyone feel nervous or left out. It’s not about scaring people; it’s about keeping the workspace honest.
Medical monitoring often gets shrugged off, especially when budgets run tight. In industries where methyl methacrylate flows daily, regular health checks spot problems before they spiral. If someone coughs more after clocking in, that’s a clue worth tracking down.
Lasting safety comes from a sense of shared responsibility. Leadership sets the tone. If foremen and managers wear their protection and ask the right questions, the rest of the team pays attention. Small reminders—like a dry erase board to check off each vent and safety shower—can carry more weight than endless posters. Real safety grows from people willing to watch each other's backs, not just following the rulebook for show.
Trust and practical steps go hand in hand. The result won’t just be compliance. It’ll be people heading home healthy, with all the worries left behind on the shop floor.
Methyl methacrylate finds its way into a lot of shops and labs. People use it to make everything from casts to dental products to flooring. What a lot of folks may not see right away is just how easily this chemical gets right into your system. I’ve stood in rooms where the sharp, almost sweet smell of methyl methacrylate hits before you even realize someone’s using it. Your nose knows trouble long before you see irritation on your skin or feel something off with your lungs. OSHA and NIOSH studied workers who handled this stuff. Documented cases of skin burns, asthma-like symptoms, and sometimes nerve trouble are not rare.Some people shrug off personal protection, maybe thinking they’re tough enough or that a few minutes of exposure won’t cause harm. That’s just not true. One coworker laughed off using gloves once, only to wind up with red, itchy hands by lunchtime. Others developed chronic breathing issues after regular contact. These stories stick with you and make you think twice about skipping safety gear.Methyl methacrylate can cause headaches, sore throats, and worse if you breathe it in, even for a short spell. Drop it on bare skin, and you get burning or maybe a rash that won’t quit. Eyes sting. All these risks show the real need to throw on proper gear, every single time.
For anybody stepping into a space where methyl methacrylate gets used, PPE wards off trouble. Good gear makes the difference between a safe shift and a trip to the clinic.The face and eyes take a beating from splashes and fumes. Safety glasses with side shields or full-on goggles protect against stray droplets. If mixing or pouring, a face shield blocks bigger splashes. Trust me, wiping this stuff from an eyebrow is nobody’s idea of a good day.Skin needs cover too. Lab coats, aprons, and long-sleeved shirts work best. A regular pair of latex gloves won’t do the trick. Methyl methacrylate slips right through some glove materials. Nitrile or butyl rubber gloves hold up better. I’ve seen folks use vinyl gloves and watch them break down before the shift’s over. It pays to check the glove’s specs and change them out at the first sign of weakness.Going without proper ventilation spells trouble fast. Fume hoods help suck up vapor before reaching your lungs. If a project means lots of fumes and not much airflow, a respirator with organic vapor cartridges gives real protection. Disposable dust masks offer nothing here.
Many employers throw PPE in a drawer and call it a day. Smart workplaces go further. I’ve worked jobs where safety meetings felt like box-ticking exercises, and nobody checked whether workers used the right gloves or masks. Real change shows up when supervisors model good habits and coworkers call out unsafe shortcuts.Good training spells out why each piece of PPE matters. I once saw a hands-on session where workers tested different gloves; only after cracking a few pairs did the lesson sink in. Clear rules about changing out contaminated gear, along with regular air checks in busy rooms, reinforce safe habits.Companies should build a routine for checking equipment, stocking quality gear, and updating safety rules. Anytime someone sees a shortcut in handling methyl methacrylate, everyone faces risk. Mistakes multiply if folks cut corners. Safety needs to feel as familiar as clocking in or grabbing a coffee—part of daily work, not just a rule on a page.Every worker has a story about the day safety gear made all the difference. Trust those stories. With methyl methacrylate, the right PPE isn’t an extra step. It’s the main one that keeps everyone on the job and out of the ER.