Nothing in the chemical world pops up out of nowhere. Chemists who worked through the 20th century learned how to pull new substances out of earlier discoveries, trying to meet new manufacturing demands and keep pace with changing ideas about safety and efficiency. Propylene glycol methyl ether acetate, known by its mouthful of a name but often called PGMEA, grew from that search for better solvents. Decades ago, traditional industrial solvents didn't pair well with safety or environmental needs. Regulations tightened, and industries like electronics, coatings, and printing hunted for something versatile. PGMEA began showing up in labs and factories, bridging the gap between performance and cleaner chemistry. The climb from the drawing board to global use wasn’t instant. Manufacturers kept tweaking production, working out the solvent’s quirks and uses, laying the foundation for PGMEA’s durable spot in the supply chain.
PGMEA runs as a colorless, nearly odorless liquid, clear enough to inspire trust but complex enough to baffle a casual observer. Its chemical backbone, with an ether and an ester group, helps it slip seamlessly into roles that traditional solvents struggled with. PGMEA dissolves a long list of resins, taking on everything from lacquers to electronics fabrication. Chemists count on its balance, letting them blend, thin, and clean without the usual headaches or safety debates. The ether-ester design makes PGMEA less volatile compared to older solvents and less likely to evaporate in the blink of an eye. Everyone in labs and factories who’s handled strong-smelling chemicals learns to value something that keeps hazards in check and doesn’t clear a room with fumes.
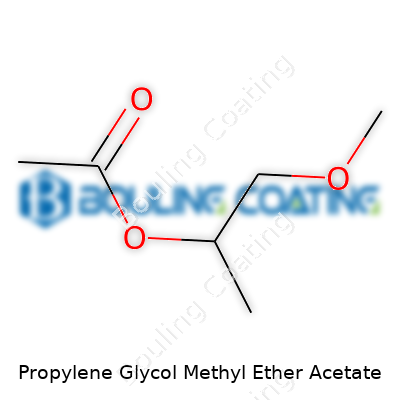
Breaking down technical details, PGMEA sits with a boiling point above 140°C and swings a low freezing point, so it stays liquid even when things get cold. Its moderate vapor pressure means workers won’t see clouds of vapor rolling through workspaces. PGMEA slips into water slowly, dissolves most resins, and brings a flash point that allows safe storage outside of specialized facilities. The molecule’s combination—an acetate group fused to a propylene glycol methyl ether—gives the liquid a knack for picking up both polar and nonpolar molecules. I’ve watched teams switch away from harsher solvents, breathing easier once PGMEA entered the mix. That minor shift in molecular structure, away from pure esters or glycols, makes a big leap when safety and usefulness count.
PGMEA sits at the crossroads of reactivity and stability, a point not many chemicals reach without drawbacks. Its slow evaporation rate becomes gold in paint and coating work, where open time fights against sagging and poor leveling. Industry insiders like to talk up how PGMEA carries pigment and gives even distribution across surfaces, leaving finishes smooth with fewer runs and streaks. In electronics, where cleaning micro-scale residues matter, PGMEA’s lower toxicity earns points. The ease of mixing with both water and organic liquids cuts down on the number of chemicals workers need to stock, making life simpler in the back rooms. From direct experience, switching to PGMEA in cleaning or thinning jobs keeps clerks and technicians out of trouble with compliance inspectors, as emissions of volatile organic compounds shrink.
Technical labels on PGMEA bottles spell out density, purity, and allowable water content. Industry standards run tighter than a drum, putting caps on impurities because even a trace can mess up delicate processes in semiconductors or paints. Those numbers aren’t just for chemists in lab coats; they protect workers from random reactions or degraded products. There’s plenty of pressure to keep quality up, especially as industries chase thinner, more responsive coatings and whiter, cleaner prints. I’ve watched QA inspectors dig into those specs, chasing down any drums outside the marked range and nipping future issues in the bud. Uniform labeling, with clear hazard warnings, sets a baseline for trust and keeps the chain of handling transparent from one stage to the next.
PGMEA comes to life through careful reactions involving propylene oxide and methanol, followed by esterification with acetic acid. It’s a process that relies on catalysts and close control of temperature and mixing rates. Getting it wrong, even by a little, leaves unreacted byproducts that risk safety or gum up machinery. Chemical engineers—often unsung heroes—run these lines balancing cost, yield, and purity. A mistake here isn’t just a problem for the numbers; it could grind entire production lines to a halt. In every factory I’ve visited, teams pay close attention to distillation and purification, aiming for a standard that keeps repeat clients coming back. Improvement cycles don’t stand still either. Plants switch out old catalysts for better ones, lean into recycling of waste, and push to cut emissions so that the process brings benefits without the baggage of outdated chemistry.
PGMEA sits at a crossroads, able to open doors to new reactions because of its hybrid structure. The molecule can undergo hydrolysis in acidic or basic conditions to split back into its component ether and acetic acid. Modifying PGMEA, such as by altering the ether or ester groups, lets chemists fine-tune volatility, solvency, and toxicity. A research group I worked with once tried swapping the acetate for longer chains, just to hunt for a blend that dried even slower in humid climates. The direct reactivity isn’t PGMEA’s biggest claim to fame, though—it’s usually the role as a solvent for something flashier. Still, the options for modification open up research and development to tailor the molecule for very specific uses, giving materials scientists a solid launching pad.
PGMEA doesn’t show up on every container label spelled out in full. Names like 1-Methoxy-2-propyl acetate, PMA, or just propylene glycol monomethyl ether acetate make the rounds depending on the supplier or the regulatory filing. This patchwork adds a challenge for workers who need to compare material safety data across borders. From first-hand experience, the confusion in labels causes slowdowns in cross-border shipping and regulatory checks. A slip in recognizing synonyms might lead to using the wrong handling procedures, upping the odds for mishaps. Each manufacturer takes pains to mark containers with the major recognized names, but there’s little excuse left for missing the hazards or the recommended protections. Information-sharing through industry groups pushes toward a clearer, more unified naming system.
Hands-on safety and handling don’t sit as optional frills with PGMEA. Workers need to use well-ventilated areas, wear gloves, and eye protection, and keep the chemical away from heat or sparks due to its flammability. Spills can pose slip hazards or risk toxic exposure if not cleaned thoroughly. Crews receive training to avoid inhaling vapors since high doses might affect the nervous system or cause headaches. Regulatory agencies, from OSHA in the US to REACH in Europe, publish strict exposure limits. Regular inspections spot-check for leaks, proper storage, and labeling. In my own experience, having clearly written checklists and walk-throughs keeps teams from taking shortcuts that could lead to costly mistakes or health scares down the line. Real-world safety means more than a binder—it’s a set of habits maintained day by day.
PGMEA’s value shines brightest in areas that demand both technical reliability and safer profiles. In semiconductor fabrication, PGMEA cleans photoresist residues from wafers at scales smaller than the width of a human hair—an operation where even tiny contaminants spell failure. Paints and coatings use PGMEA for its gentle, controlled evaporation, so surfaces dry evenly, holding color fastness and gloss. Printing inks find its solvency ideal, letting presses run faster without jamming. I’ve seen how switching to PGMEA keeps emissions and unpleasant odors down in workshops, a real breakthrough for anyone with respiratory worries. Other uses spread from adhesives in construction to cleaning agents for precision mechanics. Each application taps into a bit of the molecule’s versatility, making it hard to pin down just one “best” use.
Scientists and engineers keep testing PGMEA’s edges—pushing it into greener blends, exploring biobased routes for synthesis, and studying its interactions with new resins and substrates. Demand for low-emission, high-performance formulas guides much of this work. Recent research focuses on reducing worker exposure, improving recyclability of the solvent from waste streams, and mapping minute impurities that might disrupt sensitive technologies. Academic labs trace the life cycle, nudging manufacturers toward closed-loop production and recovery. From my time collaborating with environmental engineers, I’ve noticed research cycles shifting toward circular models, where every drop of PGMEA gets tracked and recaptured. That attention to detail brings both cost savings and tighter environmental control.
Toxicologists have put PGMEA under the microscope, running tests for short and long-term health impacts. At low exposure, most workers report mild irritation or headaches, but higher doses—often with poor ventilation or repeated spills—raise risks for liver or kidney troubles. Regulatory science entered the mix, setting occupational exposure limits and requiring companies to use fume hoods and good air exchange. Repeated studies show that managed right, PGMEA offers a much-improved profile compared to the solvents it replaced. I’ve listened to shop floor workers thank management for switching out old, harsher chemicals, reporting fewer complaints and doctor visits. Still, some unanswered questions about chronic low-level exposure hang in the air, making ongoing research and monitoring crucial in the years ahead.
Chemists and industry planners look at PGMEA as a stepping stone—an improvement but not the endpoint in the march toward safer, cleaner, and smarter chemicals. Biobased alternatives and even more effective solvent blends rest on the horizon as scientists aim higher. Regulation will keep driving innovation. Market expectations for sustainability fuel the hunt for closed-loop systems that recycle PGMEA or replace it altogether with non-toxic, biodegradable options. For factories that rely on PGMEA, continued investment in employee training, air handling, and spill response pays off in health and productivity. Each year, more of the supply chain leans on digital tracking and safer packaging. I’ve seen entire product lines reshaped to fit new environmental rules or to meet green certification. As future prospects unfold, every improvement adds up—each step shifting the legacy of industrial chemistry onto firmer, steadier ground.
Propylene Glycol Methyl Ether Acetate, or PGMEA, sounds like something for a lab coat and goggles. I remember the first time I stumbled across the name on a paint can in my father’s garage, barely able to pronounce it, let alone imagine its impact on industries and regular life. With a name like that, it’s easy to overlook how much PGMEA shapes the stuff around us.
People around the world use PGMEA in paints, coatings, and inks without giving it a second thought. Walk down any street and you’re likely passing buildings protected year-round by paints that rely on this solvent to spread evenly and dry just right. PGMEA manages to thin paints without turning them watery or changing their color — the quality matters, especially for car finishes or industrial equipment where the bare metal would rust away without that sturdy outer layer.
Anyone who’s tried to clean up paint splatters at home knows that water rarely does the trick with oil-based or acrylic paints. In these products, PGMEA helps lift and dissolve pigments and resins, making application and cleanup a lot easier. This role in cleaning up after painting or printing is no small thing for small businesses or busy families trying to keep things neat.
When people picture electronics manufacturing, they might imagine neat rows of circuit boards. What’s less obvious is the chemical dance that happens every time a semiconductor comes together. PGMEA works behind the scenes here: it helps remove photoresist in chip making and cleans up surfaces so circuits can work without short circuits or unwanted grime.
It’s easy to overlook chips when scrolling through a phone or using a computer, but without solvents like PGMEA, the miniaturization and reliability that power our digital lives would look quite different. Cleaner chips mean fewer defects, less waste, and longer-lasting devices—something we all benefit from, even if we don’t see it.
There’s always a risk when chemicals leave the factory and travel into stores, workshops, or homes. PGMEA evaporates fairly quickly, which means it can enter the air during use or disposal. Workers exposed to it in large amounts—especially indoors—may deal with headaches, dizziness, or skin irritation. Factories and regulators keep tabs on these risks, but I’ve seen plenty of DIYers paint a room with nothing but a cracked window for ventilation.
Environmental concerns matter too. Waste streams from manufacturers, paint shops, and printers might carry PGMEA into water systems or landfills. Building better ways to capture and recycle solvents from cleaning or coating processes can cut down on risks to air and groundwater. Some companies already do this with recovery systems or greener alternatives. Supporting those efforts through good policy, investment, and education can move us forward.
Most people rarely pause to wonder about the compounds in a paint can or a printer’s ink cartridge. Getting to know substances like PGMEA isn’t about memorizing chemical formulas—it’s about thinking through how products get made, how they last, and what happens after we’re done with them. Elevating the conversation about solvents and safety doesn’t just help workers or regulators. It offers everyone a better shot at safer choices and trust in what’s around us.
Chemical names usually just make most folks’ eyes glaze over, and Propylene Glycol Methyl Ether Acetate (PGMEA) sure isn’t an exception. Practically, you see this stuff floating around in everything from printing inks to paints, cleaners, and electronics. Many people at home or work have probably come across PGMEA without giving it much thought. I’ve poked around enough factory floors and workshops over the years to bump into solvents like this all the time, especially in areas where paint thinners or industrial coatings are on the shelf.
Now, the idea of "hazardous to health" deserves some straight talk. Lots of chemicals could be hazardous, depending on how they’re handled. PGMEA gives off a strong, sharp odor—pretty hard to miss in a cramped space. Catching a whiff once in a blue moon probably won’t make you sick, but breathing it on a daily basis? That’s asking for headaches, dizziness, or irritation to eyes and nose. The people I know who used to work regularly with these solvents without solid ventilation would get nosebleeds or a scratchy throat, sometimes worse if they ditched gloves and just dunked their hands straight in.
Data from occupational health studies over the years shows prolonged exposure to solvents like PGMEA raises health risks. Dizziness, drowsiness, or even trouble with coordination can creep in with high vapor concentrations. Longer-term contact with skin can dry it out or even provoke allergic reactions. Some scientific research—across countries—puts a spotlight on the need to control workplace air quality and proper personal protective gear. Nobody wants to learn about these problems the hard way.
What about the bigger picture, like whether PGMEA causes cancer or major organ trouble? So far, it doesn’t appear on lists of substances most known to cause cancer or reproductive problems, based on organizations like the International Agency for Research on Cancer and official government hazard sheets. That said, plenty of people have caught on to the wider problem: chronic exposure to mixtures of solvents in industrial jobs does take a toll on workers' health over time, even if each individual ingredient isn’t singled out as a top risk.
Communities near factories or disposal sites get another layer of concern. Solvents like PGMEA can enter the air, soil, and water, especially without strict controls in place. Even low levels in the environment bother folks with asthma or chemical sensitivities. These environmental and public health concerns usually nudge companies and cities to review their safety measures, especially as new research rolls in.
Practical steps make a difference—use gloves and goggles, ensure fans or proper ventilation draw out fumes, and substitute less hazardous solvents when possible. This isn’t just a matter for massive companies or science labs. Small workshops, print shops, or even home DIYers dealing with art supplies or old electronics ought to check the products they use. Label reading and quick checks of safety data online can go a long way.
Clear labeling, accessible safety data, and better education across workplaces have improved over the past decade or so, thanks to stronger regulations and more informed consumers. Still, accidents and exposure keep popping up, showing gaps between policy and practice. Everyone from engineers to janitors benefits when business owners walk the walk, not just talk the talk, about workplace safety.
Every time someone handles a hazardous chemical, a chain of decisions gets made, some big, some small. From where to place it once it enters a facility, to how tightly the lid gets twisted, those choices either keep people safe or put them at risk. I remember walking into a cluttered storage room at a small factory where an open bottle of acid sat next to a bag of cleaning powder. Smelling the sharp tang in the air, even a non-chemist could guess that's trouble waiting to happen. Over the years, I’ve found that common mistakes rarely come from malice—people just get too comfortable or in a rush. But comfort doesn’t protect anyone from danger when it comes to chemicals.
Some folks assume locking up chemicals is enough. From what I’ve seen, that approach lets complacency take the wheel. If a substance can burn skin, eat through metal, or poison air, storing it in any old cabinet turns a small risk into a big gamble. The temperature in the storage area often makes a real difference. Many chemicals break down or react if they get too warm or freeze in cold weather. In my own work, even a few degrees off made plastic containers brittle, allowing contents to leak and cause stains that took weeks to clean up. Nobody enjoys dealing with a mystery spill at the end of a shift.
I once helped organize a community cleanup, where we found several bottles labeled “solvent” and “blue stuff” in a garage. There was no way to tell what was inside, and mixing the wrong materials can bring disaster. Relying on memory or “we always put this here” never beats clear, up-to-date labeling. Imagine trying to explain leftovers to a roommate—nobody wants to play guessing games, especially with something that could hurt them. At work, the solution comes down to consistent labeling and sticking to it. Faded labels can put emergency crews, coworkers, or anybody else who stumbles onto that shelf in harm’s way.
Years ago, at a job site, a coworker ignored glove recommendations while handling a caustic cleaner. He thought a towel would be enough. Five minutes later, pain told him differently. Whether for acids, solvents, or even “inert” powders, protective gear fits more than just regulations; it protects real people. Shops and labs with regular, hands-on training sessions see far fewer accidents than those relying only on posters and thick binders. Familiarity does not mean safety, especially with chemicals that don’t look or smell dangerous until it’s too late.
Precautions should not end at strict rules. OSHA data shows that proper separation of incompatible materials prevents hundreds of hospitalizations each year. Separating oxidizers from organics or acids from bases—simple steps, but too easy to skip on a busy day. Encouraging every worker to point out short-cuts without fear of backlash can create a real culture of safety. After all, nobody pays the price of neglect quicker than those closest to the spill or the exposure.
We can take a page from shops and labs with tight routines—rotating stock so older product gets used first, checking seals and dates regularly, using spill trays and ventilation where chemicals are transferred or opened. Solutions don’t cost a fortune but skipping them can cost lives, health, or livelihoods. Urgency and convenience push people to cut corners. Reminding ourselves and our coworkers why the rules exist every day keeps everyone going home safe at the end of a shift.
Many folks outside the industrial or chemical circles probably won’t recognize Propylene Glycol Methyl Ether Acetate—often abbreviated as PGMEA. Its chemical formula C6H12O3 and CAS number 108-65-6 tend to appear mostly on technical safety documents and regulatory paperwork. But PGMEA quietly supports a surprising number of everyday products, drilling far beyond simple statistics and lists. Having spent years tracking chemical safety in workplace settings, I frequently saw this compound crop up. If you want to understand why manufacturers and engineers pay such close attention to PGMEA, you don’t need to squint at obscure chemistry charts—you only need to look at the big impact that small molecules can have.
Shopping for paint, ink, or electronic components? There’s a high chance PGMEA helps bind, dissolve, or thin the substances inside. This solvent is everywhere from high-tech chip plants to the art supplies on your desk. When buyers and users see that long chemical name, the formula and CAS number become more than bureaucratic details. A correct identification separates safe use from dangerous mix-ups. Precision like this has real-world consequences. Tech companies rely on that unique identifier to make sure nothing hazardous or counterfeit slips into their clean rooms. The wrong solvent could mean lost time, ruined materials, or even a workplace injury. For painters, even a small substitution might mean unpredictable drying times or harmful fumes.
Although PGMEA serves as a go-to option in manufacturing, labs, and studios, it’s no stranger to health concerns. Inhalation brings headaches, dizziness, and over longer exposures, possible effects on the liver. Even brief contact can irritate skin or eyes. My own field observations taught me that too many workers still reach for gloves and masks only after experiencing symptoms. Regulators in North America, Europe, and Asia cite PGMEA frequently in workplace safety advisories, arguing it deserves proper handling each time it’s used.
So what makes the formula and CAS number so important in daily practice? These aren’t just codes on a package or bottle—they connect back to a web of safety data, research, and guidelines. Employers find it easier to create protocols, detect leaks, and avoid harm. Workers feel more secure identifying exactly what’s in front of them, not just relying on product labels or marketing promises.
Wider training in chemical literacy stands as the most practical way forward. That means don’t just list formulas, but explain their meaning. A safety meeting that highlights the health effects and regulatory signals attached to PGMEA makes the compound real—not something you glaze over. Facilities would do well to post CAS numbers with clear hazard signage where PGMEA is handled or stored. Suppliers investing in better education for buyers and users can also reduce tragic mistakes.
The world behind C6H12O3 and 108-65-6 goes beyond formulas and numbers. It touches product quality, workplace health, and consumer trust. The conversation needs to keep connecting technical precision with real outcomes, so everyday choices about “what’s in this bottle” or “is this safe for my job” become more straightforward and less risky—for everyone, from line workers to families at home.
The real world doesn’t treat chemicals like harmless puddles, and Propylene Glycol Methyl Ether Acetate—PGMEA for short—works as a reminder. This solvent shows up in paint thinners, printer ink, and electronics manufacturing. It slides its way into production lines, often without drawing much attention. But spills or careless wicks across a glove turn into headaches of their own, for both people and the place they work in.
PGMEA does more than irritate skin. Breathing in its vapor twists up your respiratory tract and even light exposure leaves you light-headed. The risk pounces faster in tight workrooms or places with little fresh air. From what I’ve seen, people dismiss the small stuff, until the headache hits or someone’s trying not to cough. Factory life runs on practical lessons, not just rules posted by the door.
Symptoms tell a story. Stinging eyes, raw throats, or feeling faint after a spell near a leaky drum. These aren’t quirks—they’re calls to stop and think. A label or data sheet can help, but crews trust their own eyes and noses more. We can’t expect a safety manager to catch every puddle. Each person on the floor holds the next one’s health in their hands.
Stopping a spill means moving fast, but not without a plan. Plenty of workers get hurt not during the spill, but during the scramble to clean it up. So the first job: clear folks out of the area, kill any ignition sources, and throw open some windows if possible. A buddy once told me, “If you can smell it, you’re too close without a mask.” I’ve seen that proven right, time and time again.
Those paper towels sitting in the break room will just spread the mess around. You need proper absorbents designed for solvents—think pads, not wads of napkins. No open skin, ever, so gloves rated for chemicals and a pair of decent goggles save more trouble than most realize. After scooping up the worst of it, containers built for hazardous waste come out. Some toss wipes in the trash and hope for the best; I’ve seen that nearly spark a fire later on.
Relying only on the folks with fancy titles or hazmat training just invites trouble. Experience tells me that a team’s level of care goes up once they know spills can happen to anyone, and fast. It’s still too common to see gloves tossed on the ground, or broken containers set aside because “someone else will deal with it.” Shortcuts bite you back—either with health issues or fines that eat into budgets better spent elsewhere.
Every company brags about safety, but only a handful keep spill kits where they’ll really be used. Training drills sometimes feel pointless until the real thing jumps up and disrupts a shift. Having clear steps written in plain language, not just safety-speak, helps cut through panic. Regular walk-throughs and honest conversations work better than posters that fade with time and dirt. In practice, workers who’ve had the right training approach spills with the same focus they give to their most important equipment.
We may not see every risk, but ignoring what we do know—the bite of PGMEA, the price of complacency—costs more than just a stained pair of boots.