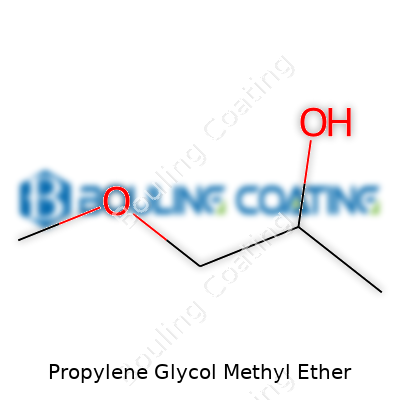
Decades ago, the chemical sector, thirsting for safer alternatives to harsher solvents, introduced propylene glycol methyl ether, often called PM or PGME. Seeing industries transition from more volatile, dangerous compounds to solutions that both work and pose fewer health hazards matches the narrative that runs through advances in chemistry—solving problems that used to seem insurmountable. In the 1980s and 1990s, PM carved out its spot in paints, inks, and cleaning products. Back then, people chased performance, even at a cost to safety. Standards started to shift as more researchers and end-users demanded compounds that balanced both workability and safety. That change, incremental but real, owes a lot to the emergence of solvents like PM.
Propylene glycol methyl ether offers something both chemists and manufacturers respect: versatility with a manageable profile. Walk through a paint shop, printing facility, or electronics plant, and odds are strong you’ll spot PM in use. What stands out is its ability to dissolve greases, resins, and dyes without tearing through equipment or releasing clouds of dangerous fumes. It boils well below the temperature of water—a fact that makes it a favorite where volatility matters, but safety can’t be put on the back burner. The clear liquid doesn’t bring an overpowering odor to the workplace, making respirators and ventilation systems a little less critical compared with older solvents.
From hands-on experience mixing inks and coatings, the characteristics of PM contribute to smooth blends and consistent quality. It keeps blends stable, thanks to its mid-range evaporation rate and strong solvency. Technicians often appreciate how it removes sticky residues without leaving streaks behind—something that isn't just theoretical but proven on factory floors. Chemists point out the relatively low toxicity and decent compatibility with water, making it easier to handle during both blending and cleanup. Those properties don’t just sit on a data sheet—they translate into shifts in workflow, less downtime, fewer complaints from workers, and less maintenance on air filtration units.
Labels tell part of the story—CAS number, chemical formula, boiling range, and flash point. From a regulatory point of view, those numbers drive crucial decisions about storage, shipping, and workplace protections. Real-world work goes deeper, focusing on how slight impurities affect the end product or how mix ratios shift under different temperatures. The move toward stricter requirements for purity and traceability has forced suppliers to adjust their own production lines and tighten documentation. That extra vigilance isn’t just for ticking boxes; it helps cut down on product recalls and workplace exposures.
The process for making PM involves reacting propylene oxide with methanol, usually with careful control to capture as much of the preferred isomer as possible. At industrial scale, the process demands just the right catalysts and operating conditions, or efficiency suffers. I've seen how tweaks in reaction temperatures or feed purity can shift output dramatically. These decisions ripple out to workers handling the stock and to plant managers balancing waste and yield. Emissions controls have become stricter over the years, and the drive to minimize leftovers and byproducts nudges companies toward greener, more contained methods.
In practice, PM doesn't just stand on its own—it often gets mixed or reacted to tailor performance. Chemists modify it by etherification or further functionalization to nudge solvency, boiling point, or even skin absorption. That lets product developers tune cleaner blends or thinners for tasks as varied as degreasing engines and printing fine graphics. Looking at product trends over the last decade, adaptability in PM-based blends has opened up opportunities to replace nastier solvents. In the field, innovation rests on incremental tweaks, not flashy overhauls.
Depending on market and context, propylene glycol methyl ether goes by a handful of aliases. Propylene glycol monomethyl ether, PGME, 1-methoxy-2-propanol—they all track back to the same core compound, though specific applications sometimes demand one isomer or grade over another. For anyone moving between regions or reading safety data sheets, the changing names prove more than a paperwork headache—they can run companies afoul of regulations if labels and documentation fail to track the right synonyms.
Every industry that relies on PM balances ease of use with responsibility. Guidelines target exposure limits, local exhaust systems, proper gloves, and splash goggles—not just officially, but because peer experiences bear out their importance. In labs, old-timers walk new hires through spill controls and eye washes, emphasizing why safety routines matter. Even with relatively low acute toxicity, long-term exposure without adequate protection still brings risks, including skin and eye irritation or headaches. Regulatory updates in the last few years reflect pressure from labor groups and health studies, pushing toward further cuts in allowed exposures and stricter accident reporting.
Applications for PM span more than just solvent roles in paints and inks. Electronics manufacturers tap it for cleaning sensitive circuitry. Pharmaceuticals appreciate its balance of solvency and safety, particularly for topical or aerosol preparations that can’t tolerate harsh residues. In graffiti removers, PM does the heavy lifting that less powerful solvents can’t handle. Knowledge passed down between workers often focuses on those “trouble spots” where stronger solvents would damage plastics or coatings; PM gets the nod because it solves without destroying. As industry demand keeps growing, especially for green-certified and low-toxicity ingredients, PM keeps drawing attention.
Development around PM—both on the basic and applied science sides—keeps moving. University labs and industrial R&D wings look for ways to deepen PM’s solvency, cut emissions, and trim energy use in production. Researchers invest time in tuning the isomer mix, switching to renewables for feedstocks, or blending PM with co-solvents to serve niche markets. Experience shows that time and resources spent on cleaner, less toxic modifications pay off through lower regulatory risk and stronger long-term sales. New analytical tools let teams spot impurities at lower concentrations, letting quality assurance teams deliver more consistent product to end users.
Decades of animal studies and workplace surveys have shaped our understanding of PM’s health profile. Acute toxicity registers as low, but chronic exposure can still cause headaches, drowsiness, or skin dryness. I’ve seen how scattered attention to proper controls in small shops or print houses leads to subtle but important discomforts among employees: irritated eyes, slow reaction times, or mild nausea. Toxicologists highlight metabolites that clear pretty quickly from most healthy bodies, but the margin for error shrinks in poorly ventilated spaces or with high loads. Recent studies keep a cautious tone, emphasizing the need for monitoring, especially for vulnerable or pregnant workers.
Demand for PM reflects shifts in manufacturing, consumer safety expectations, and green chemistry standards. Regulatory bodies are pushing for safer substitutes even for compounds that seem relatively tame. Investors and industry planners look for candidates that maintain or boost performance without trading away workplace or environmental safety. Over the long haul, continued innovation in synthesis—particularly methods that drop fossil-based inputs—could reshape how PM is made and used. In my view, the most promising future for PM lies in refining its production to lessen environmental burdens, digging deeper into blend customization, and responding rapidly to new research on chronic exposure. The focus isn’t just on making things work—it’s on making them safer, smarter, and kinder to both people and the planet.
Whenever I try to explain what propylene glycol methyl ether—let’s call it PGME—does, I picture an invisible workhorse. Most people walk by its impact without noticing. But in paint shops, printing rooms, and even janitorial closets, PGME shows up in ways most folks can’t see. This chemical has its fingerprints all over tasks that make life a bit simpler or more colorful.
The street-level truth about PGME is its role as a solvent. Companies turn to it to thin paints, dissolve inks, and help glues stick just right. Anyone who’s painted a room probably owes the smooth application, in part, to PGME hiding in the can. I’ve stripped years-old stickers off glass using household products without knowing this chemical helped the solvent lift off gunk without a struggle.
Follow a magazine from printing press to coffee table, and PGME has likely played a part. Inks come out vibrant and sharp partly because this solvent lets pigments flow evenly across paper. Over in cleaning products, folks using glass cleaners or degreasers run into the chemical too. It cuts through greasy fingerprints and sticky residues faster than old-school soaps. Factories and workshops trust it for speed and efficiency, getting machines back online in less time.
The more industries use PGME, the more questions show up about safety. You’ll see it listed on product safety data sheets. Direct skin contact or fumes from concentrated forms can irritate your hands or lungs, so masks, gloves, and decent ventilation matter. Having spent time in print shops and auto body shops in my younger days, I learned quick that even everyday solvents deserve respect. Locked cabinets, clear labels, and proper training kept workplace accidents rare, but that diligence makes a huge difference.
Rules about volatile organic compounds (VOCs) push manufacturers to rethink recipes for paint and other formulas. Too many VOCs released into the air can worsen smog and ground-level ozone. PGME counts as a VOC, so companies have had to balance a cleaner environment with getting the job done. As cities try to tackle air pollution and indoor air quality, finding safer alternatives becomes less of a trend and more of a necessity.
Not every replacement for PGME works the same way, and shortcuts don’t always pay off. Water-based paints sound great but changing a formula can require completely new equipment. Some cleaners promise lower emissions, but users often complain the results don’t match what they got with PGME-based options. Right now, research focuses on solvents that keep workers and customers safer while meeting stricter environmental targets. I’ve seen changes take root best when companies bring workers into the process—not just telling them what’s different, but showing why it matters. Better substitutes will take creative chemistry, open communication, and customer feedback that moves companies to keep raising the safety bar.
PGME still has a seat at the table in many industries, but the drive for safer and greener chemistry grows stronger each year. The story of this solvent runs deeper than any label or fact sheet, because it intertwines with cleaner air, safer jobs, and how we approach the invisible helpers that keep the world moving.
People ask about chemicals in daily products all the time, and propylene glycol methyl ether (PGME) often pops up when the conversation turns toward paint thinners, cleaning fluids, or industrial manufacturing. PGME handles jobs like dissolving tough residues or thinning out paints, which puts it in the toolkits of painters, janitors, and factory workers every day. Safety matters here—not just for the skin, but for the lungs and the wider environment. It's worth getting past the technical labels and figuring out what real risks show up in practical, real-world use.
I’ve walked through workshops and paint booths where PGME floats through the air, mixed into other fumes. Breathing it in, even for a short time, can bring on a headache, cough, or sore throat. Extended exposure ramps up irritation—sometimes leading to dizziness or feeling lightheaded in a hot, stuffy room. Studies from toxicology centers pin most issues on inhalation. Once airborne, PGME pushes right into the bloodstream through the lungs, and unless proper ventilation works overtime, people might suffer more than just an upset stomach. Skin contact draws less attention, but it can dry or crack skin after repeated splashes. Regulatory agencies have recognized these problems for years and set limits around how much workers should breathe in during an eight-hour shift.
Most people outside of tough industrial settings won’t meet PGME in high doses, but it sneaks into some household cleaners and hobby supplies. Here, the risk drops, though accidents still happen—kids getting into cleaning bottles, someone ignoring labels and mixing products in a closed bathroom. History proves overexposure doesn’t wait for a factory setting. People who don’t understand what they’re handling sometimes end up at the doctor, and responders find the same chemical culprits as in big industry.
Inventors and manufacturers didn’t pick PGME out of a hat. This chemical does its job without making products flammable or causing hazard sheets to sound extra alarms compared with solvents like toluene. Still, no level of convenience justifies turning a blind eye. Studies from reputable groups, including the National Institute for Occupational Safety and Health, have reviewed the cumulative effects of chronic exposure. Their take: Fix ventilation, use gloves, train workers, and switch to safer chemicals if possible. It’s about making straightforward changes that pay off over a lifetime of work.
Completely erasing PGME from industries and cleaning aisles tomorrow would be tough. Still, a few good habits make a difference. Big employers who update extractor fans and supply the right protective gear cut down on health complaints among their crews. The same spirit applies at home—read product labels, crack a window, and lock chemicals out of reach from kids. Regulators already limit workplace exposures, but more public awareness matters just as much. Most risks from PGME shrink as soon as people respect it, read instructions, and act as if their health and their coworkers’ health weigh more than getting the job done fast.
Plenty of folks working in labs or factories overlook the real stakes when dealing with Propylene Glycol Methyl Ether—PGME for short—because it isn’t as notorious as some chemicals. I’ve seen accidents and near-misses that came down to small habits, things like skipping labels, storing next to something it shouldn’t touch, or just getting lax with container seals. People get comfortable, and that’s where problems start.
On paper, everyone knows PGME should stay in a cool, well-ventilated spot, away from sparks and open flames. This stuff has a low flash point, so it takes less heat than you might expect for it to catch fire. Some may stash it in a regular chemical cabinet, but I’ve run across leaky jugs and sticky residue on the shelves more often than I’d like to admit. Over time, those mistakes add up to real danger. The odor sneaks up, especially if you’re not paying attention, and flammable vapors don’t care if the last inspection was only a month ago.
I remember a small warehouse where the air conditioning broke in July. Somebody left a half-used drum near a window, and by afternoon, the room stank. One static spark could have set the whole place off. Supervisors scrambled to replace every cap and drum before someone got hurt. This close call hammered home how PGME doesn’t forgive lazy storage.
I’ve lost count of how many times spills happened during transfer simply because someone used an open container or a broken funnel. That smell clings to your clothes and stings your nose for hours. The safety goggles and gloves matter, but so does thinking two steps ahead—never pouring from large drums above shoulder height or rushing to top off a tank at the end of a shift. Bad habits like decanting just outside the ventilation hood, or propping a door open for ‘airflow’ all seem minor, but multiply the risk.
There’s also the matter of work culture. Some teams treat chemical rules like burdens, while others make safety reflexive by talking about close calls openly and not shaming mistakes. I see better adherence in places where anyone can speak up if they spot a risky shortcut. They've even taped clear “flammable” signs and lists of incompatible substances on shelves—simple, direct reminders that nobody ignores.
From experience, most storage and handling problems clear up when people stop treating flammable chemicals as routine. Locking cabinets and automatic closers help, but I’ve learned that regular walk-throughs by a knowledgeable supervisor catch more than any sign or checklist. Even just marking safe fill lines on drums and setting 'no food' zones around storage areas goes a long way. If something leaks, people will spot it faster if the area’s clean and organized.
Tighter storage practices paired with honest conversations about dumb near-misses set a tone—no one’s too experienced to skip a safety step. Storing PGME safely and handling it with focus means thinking of worst-case scenarios before they become real events. Every day, lots of small choices add up to either a safe shift or tomorrow’s accident headline.
Ask anyone working in painting, cleaning, or laboratory settings, and the name Propylene Glycol Methyl Ether pops up fast. People often talk about what it does or where it fits in, but fewer pay close attention to what makes this liquid tick. For many, it’s a clear, faintly sweet-smelling solvent that moves through supply chains quietly, doing serious work in the background. I’ve spent enough time around labs and warehouses to know it’s rarely the star of the show, yet it deserves a sharper look.
Pour a bit of Propylene Glycol Methyl Ether, and it spreads out fast. It doesn’t cling to skin as viscously as some glycols do. Touch reveals a liquid that feels almost thin, hardly greasy. The boiling point tips past 100°C, making it useful in processes where you don’t want everything flashing off early. Unlike old-school, high-toxicity solvents, it isn’t as immediately flammable as acetone or benzene. That’s a peace of mind value you can’t fake in small shops or big factories alike. No one wants to worry about intense vapors building up with every brushstroke or mop.
On a hot day, you’ll notice the vapor rising, but the scent never zeros in on your sinuses like some other chemicals. In my experience, this soft approach makes it easier to work with during long jobs. Rinsed off glassware dries quickly without residue, meaning it clears the stage for the next round without fuss or extra scrubbing.
This ether’s chemical side matters to plenty of trades, from formulators mixing up coatings to cleaners developing streak-free products. It blends easily with water and many other organic solvents. In a shelf full of aggressive or delicate chemicals, this flexibility saves headaches. Folks making water-based paints get a smoothing hand; it lets pigments and binders spread without the sludge and streak of poorly chosen solvents.
Its structure brings a measure of stability, resisting quick breakdown when combined with acids or bases, so it doesn’t morph into something dangerous without a strong push. That reliability counts. Across my work, people trust substances that don’t surprise them halfway through a shift.
Experience shows that physical and chemical traits speak louder than labels. A balance between drying time and safety changes how, where, and by whom a chemical gets used. Propylene Glycol Methyl Ether’s boiling point means it won’t quit early on big surfaces. The moderate evaporation rate helps keep costs in check, since you lose less to the air. Labs avoid frequent top-ups, painters see fewer dried brush marks, and cleaning crews escape headaches from harsh odors.
Its compatibility with water-based formulations signals a better environmental profile than the heavy hitters of the last century, which pollute air and water on a massive scale. Looking at studies and workplace stories, even its relatively low human toxicity, compared to methylene chloride or toluene, removes a layer of risk for workers who handle chemicals daily. While no one should skip gloves and goggles, people breathe a bit easier—literally—knowing they aren’t dodging as many health problems over time.
The story of Propylene Glycol Methyl Ether calls for transparency and practical thinking in chemical use. Instead of chasing the next miracle solvent, industry leaders need to invest in smarter ventilation, upgrade protective gear, and swap out old-solvent habits. Regulators, too, can demand clearer safety data and limit harmful exposures, especially for small-business crews often left out of big safety conversations.
Lately, green chemistry is catching up, encouraging research that keeps function strong while raising the bar for safety. Propylene Glycol Methyl Ether stands as an example; it proves that common-sense design—balancing task performance with fewer health worries—can carry real weight wherever work gets done.
Too often, people treat chemical waste as if it’s no different from dirty dishwater. That’s not just careless—it's one way that toxins slip into streams and drinking water. Propylene Glycol Methyl Ether (PGME) doesn’t get the same headlines as oil spills or pesticides, but the risk to health and ecosystems is real if we take shortcuts.
This solvent finds its way into paints, inks, and cleaners, working quietly behind the scenes. After years in the coatings industry, I saw firsthand how a casual attitude toward disposal can spell trouble. A single flush of chemicals might seem harmless, but repeated carelessness builds up. Legally and ethically, it’s on us to keep these compounds out of the water system.
Too many people assume that anything liquid can go down the sink or storm drain. That couldn’t be further from the truth. Wastewater plants aren’t designed to scrub out every solvent or compound. PGME can slip through, ending up in rivers or groundwater supplies. Fish and aquatic insects pay the price, and eventually, so do we.
Federal and state guidelines treat solvents like PGME as hazardous waste. In practice, this means collecting used solvent in clearly labeled containers. From there, a licensed waste handler takes it for high-temperature incineration or fuel blending at approved facilities. No one loves extra paperwork or fees, but the alternative is polluting our own backyard in slow motion.
Responsible disposal is easier with good habits. At one warehouse, our crew used color-coded bins, backed by regular safety briefings. It meant a little more effort, but it cut confusion and stopped accidental dumping. Shops can take advantage of local waste collection programs—often funded in part by community fees or industry groups. In some areas, hazardous waste roundup days let the public or small businesses drop off leftover chemicals for safe treatment.
Ideally, we’ll use less hazardous stuff in the first place. Some companies already experiment with greener substitutes—blends that clean up just as well but break down safely. These shifts don’t always come easy or cheap. Until the safer solutions are everywhere, we have to keep solvents like PGME out of landfills and water. That means spreading knowledge about safe handling, pressuring suppliers for less toxic options, and supporting laws that protect our water.
Safe disposal calls for more than just compliance. It’s about thinking past the short term and choosing to leave less mess for the next person. I’ve seen too many shortcuts turn into problems down the line—wells tainted, fines dealt out, and neighbors angry because someone couldn’t bother to ask where stuff should go. The right approach isn’t a secret—it just takes all of us actually caring enough to get it right.